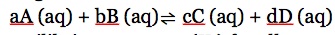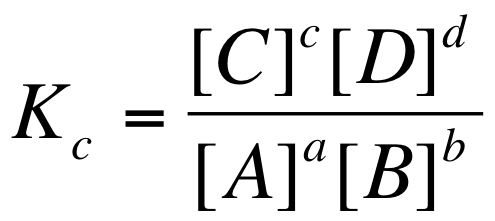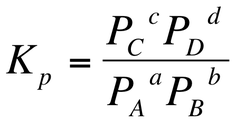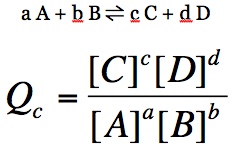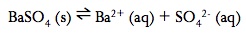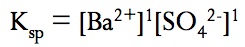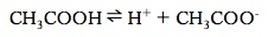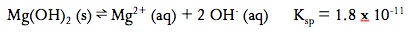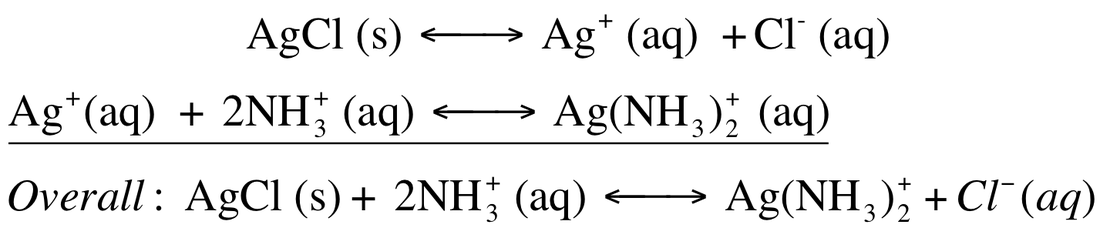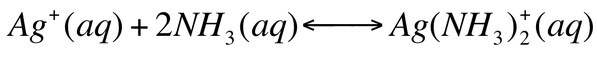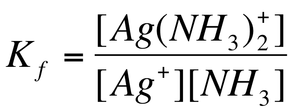Swimming - Chemical Equilibrium
|
Few sports have a wider variety of events then the sport of swimming. From the butterfly to the freestyle to the backstroke, there are many diverse events in the sport of swimming.
Just like there are diverse sports in swimming, so to are there a wide array of calculations that you can perform with equilibrium constants. From Kp, Kc, Ksp, Kf, Ka, and Kb, we have covered a lot of material on equilibrium constants. This section will cover all of the equilibrium information that we have covered except acids/bases. REMEMBER the first question on the FRQ every year is an equilibrium problem, so make sure you know this content well! |
Equilibrium Constants
At equilibrium the rate of the forward reaction is equal to the reverse reaction.
At equilibrium, the
rate of the forward and reverse reaction are equal, but continue to occur. We are at dynamic equilibrium because the reaction is occurring, but rates are equal.
The ratio of products to reactants in equilibrium is known as the equilibrium constant (K). Only written for aqueous and gaseous substances. Liquids and gases do not participate in equilibrium. For the general reaction:
The ratio of products to reactants in equilibrium is known as the equilibrium constant (K). Only written for aqueous and gaseous substances. Liquids and gases do not participate in equilibrium. For the general reaction:
The equilibrium constant (Kc) for all aqueous substances is:
When the substances in an equilibrium, we can calculate the equilibrium constant using partial pressures. This is represented as Kp. Please note that Kp≠Kc. For the equilibrium previously mentioned, if the substances has been in the gaseous state, then the equilibrium constant would be written as:
A large K indicates that there are more products than reactants at equilibrium. A small K indicates that there are more reactants than products at equilibrium. If K >> 1, then it is products favored. If K << 1, then it is reactants favored.
Adding Equilibrium Constants
Like H, when we manipulate a chemical reaction, the value of the equilibrium constant changes. Three possible changes:
1.Running Reaction in Reverse
2.Multiplying Reaction by Common Factor
3.Adding Equilibriums
1.Running Reaction in Reverse
2.Multiplying Reaction by Common Factor
3.Adding Equilibriums
ICE Tables (Kc and Kp)
—Oftentimes, we know the amount of substance that we begin with, but the equilibrium amounts are unknown.
—We use stoichiometry to help solve for the equilibrium concentrations using the initial conditions and the value of Kc.
We use ICE tables in order to calculate equilibrium concentrations. ICE Tables can be set up as follows:
—We use stoichiometry to help solve for the equilibrium concentrations using the initial conditions and the value of Kc.
We use ICE tables in order to calculate equilibrium concentrations. ICE Tables can be set up as follows:
Reaction Quotient and Le Chatlier Principle
Oftentimes, we examine reactions prior to them achieving equilibrium. How do we know the direction a reaction needs to move in order to achieve equilibrium? We use the reaction quotient (Q) to determine this. Q is calculating the equilibrium constant prior to equilibrium:
There are three possible results of our Q calculation:
- Q = K: Reaction is at equilibrium
- Q > K: Amount of products is too large, reaction needs to shift towards reactants
- Q < K: Amount of reactants is too large, reaction needs to shift towards products
When you calculate Q you are doing the math behind Le Chatelier’s Principle. Le Chatelier's Principle states that:“If a system at
equilibrium is disturbed by a change in temperature, pressure, or a component
concentration, the system will shift its equilibrium position so as to
counteract the effect of the disturbance.”
If we add more of a product/reactant, then our reaction will shift to the other side in order to compensate for this new amount.
Changes in pressure can alter the side of the reaction that is favored. If we increase pressure, the reaction will shift to the side that produces less molecules. If we decrease pressure, the reaction will shift to the side that produces more molecules.
When a reaction is endothermic, heat is listed as a reactant.
Heat + Reactants --> Products
When a reaction is exothermic, heat is listed as a product.
Reactants --> Products + Heat
Therefore we can treat heat like a reactant or a product depending where it is listed.
If we add more of a product/reactant, then our reaction will shift to the other side in order to compensate for this new amount.
Changes in pressure can alter the side of the reaction that is favored. If we increase pressure, the reaction will shift to the side that produces less molecules. If we decrease pressure, the reaction will shift to the side that produces more molecules.
When a reaction is endothermic, heat is listed as a reactant.
Heat + Reactants --> Products
When a reaction is exothermic, heat is listed as a product.
Reactants --> Products + Heat
Therefore we can treat heat like a reactant or a product depending where it is listed.
Solubility Equilibrium (Ksp)
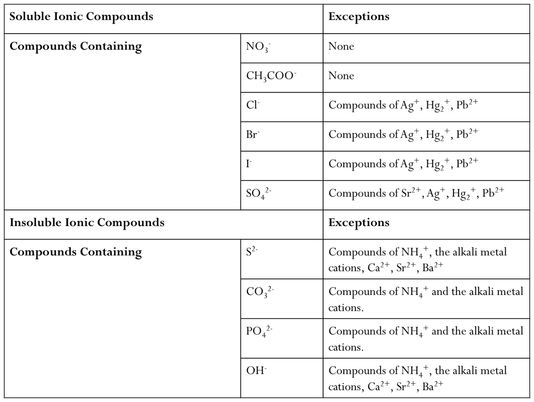
Many reactions results in the production of a solid compound know as a precipitate. These reactions are known as precipitation reactions. We can determine whether or not a solid will form based on a few rules.
A saturated solution is one that has dissolved as much solid as possible. In a saturated solution, equilibrium is established:
The equilibrium constant for slightly soluble salts is known as the solubility – product constants or Ksp
The Ksp is NOT the solubility. Solubility describes how many grams can dissolve in a liter of solution. The Ksp describes how much dissolves to form a saturated solution. Solubility can change, but Ksp CANNOT. Remember that when solving for molar concentrations in a slightly soluble salt equilibrium, the value of x represents the molar solubility or the number of moles of solid that can dissolve per liter of solution. This molar solubility can then be converted either to mass solubility (g/L) or to concentrations of the ions and/or the Ksp from your ICE table.
Impacting Equilibrium
There are 3 ways in which we can impact the solubility of a slightly soluble salt. They are:
1. The common ion effect
2. Altering the pH
3. Formation of Complex Ions
1. The common ion effect
2. Altering the pH
3. Formation of Complex Ions
The Common Ion Effect
Recall the acetate equilibrium:
If we had both acetic acid and acetate in solution, than the acetic acid will ionize less in order to achieve equilibrium. There is very little movement needs to be done in order to achieve equilibrium. Whenever a weak electrolyte and a strong electrolyte containing a common ion are together, the weak electrolyte ionizes less than when it is alone in solution. This is known as the common ion effect.
pH and the Solubility
The pH of a solution impacts the solubility for any compound that has a basic component when dissolved in solution. Consider the equilibrium:
If we are in a basic solution, then we will have more solid due to the presence of extra hydroxide ions. The solubility of a compound containing a basic component increases as the pH is lowered. The solid is more soluble in an acid due to the removal of OH from solution by an acid base reaction. This pushes our overall equilibrium to the right. Furthermore, if OH is present, then we have a common ion present and the solution will favor the formation of the solid.
Complex Ion Formation and Solubility
When transition metals bond they are the central atom and they bond extensively with numerous substances. The complexes that form are known as metal complexes. The molecule that bond with metals are known as ligands. A ligand is always a Lewis Base and the most common ligands are H2O, NH3, Cl-, and CN- due to their free lone pair of electrons that can be donated to a metal. The number of ligands bound to the metal is the coordination number.
The formation of complex ions can drastically impact the solubility of a metal. Ligands act to pull metal ions into solution and displace solvating H2O, thus, making the solution more soluble. We can make a solid, like AgCl, more soluble by adding NH3. The two equilibriums can be written as:
The formation of complex ions can drastically impact the solubility of a metal. Ligands act to pull metal ions into solution and displace solvating H2O, thus, making the solution more soluble. We can make a solid, like AgCl, more soluble by adding NH3. The two equilibriums can be written as:
For the equilibrium
We can calculate the formation constant (Kf). The formation constant is the equilibrium constant for the formation of the complex ion from the metal ion and the ligand. These are extremely large number as they usually go to completion.
Selective Precipitation
Recall, the reaction quotient (Q) can be used to determine the direction an equilibrium needs to shift in order to achieve equilibrium:
Oftentimes, we have solutions that contain multiple solubility equilibriums. We can selectively precipirate some of the compounds, but not all, by utilizing Ksp. The smaller the Ksp of an ion, the more likely it will precipitate from solution first. We can calculate the concentration that is required to precipitate by setting up an ICE table and determining which solution requires a smaller concentration of the ion being added in order to form a solid. The ion that requires the smaller amount of ion will be the one that precipitates first.
- Q > Ksp: Shift left and precipitate more
- Q < Ksp: Shift right and dissolve more
- Q = Ksp: Solution is at equilibrium
Oftentimes, we have solutions that contain multiple solubility equilibriums. We can selectively precipirate some of the compounds, but not all, by utilizing Ksp. The smaller the Ksp of an ion, the more likely it will precipitate from solution first. We can calculate the concentration that is required to precipitate by setting up an ICE table and determining which solution requires a smaller concentration of the ion being added in order to form a solid. The ion that requires the smaller amount of ion will be the one that precipitates first.
Practice, Practice, Practice
|
Once you feel you have reviewed an adequate amount of the material, try practicing a few laps with these practice problems.
|
The Final Heat!
|
Once you feel you are ready to stare down Michael Phelps and challenge him for the gold, complete the following FRQ problem. NOTE - If you need retake the FRQ, then you need to see Mr. Astor for another question.
|