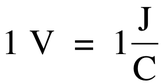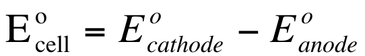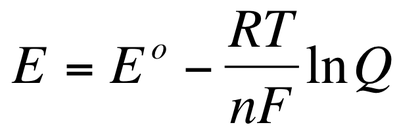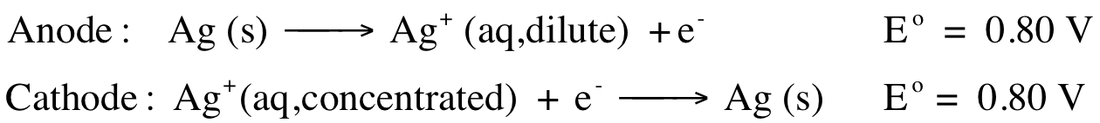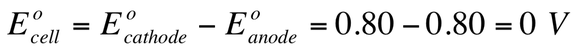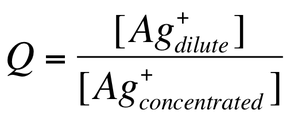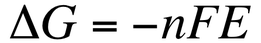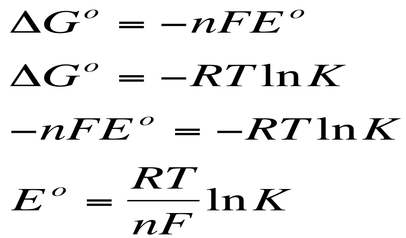Triathlon - Electrochemistry
|
Perhaps the most physically grueling event is the triathlon. A triathlete must:
1. Swim 1 mile 2. Bike 25 miles 3. Run 6.2 miles The athlete that emerges is truly one of the toughest at the olympics. Chemistry has a similar triathlon and that is electrochemistry. An expert chemist must be able to use electrochemistry to convert between 1. Gibbs Free Energy 2. Equilibrium Constants 3. Standard Electrode Potentials (Eo) If you emerge victorious from this event, then you will truly be one of the contenders to win the AP Olympics. |
How Batteries Work:
Group Project Google Doc - Make a Copy and Get Rollin!
How Batteries Work by Robert L. Wolke
Wired Magazine - Electromotive Force, Separation of Charge, Voltage
123D Circuits - Components - Circuit Scribe -
Group Project Google Doc - Make a Copy and Get Rollin!
How Batteries Work by Robert L. Wolke
Wired Magazine - Electromotive Force, Separation of Charge, Voltage
123D Circuits - Components - Circuit Scribe -
Balancing Redox Reactions
We cannot balance redox reactions the same way that we balance other reactions that we have studied. This is due to the fact that sometimes the reactions are run under acidic conditions and other times they are run under basic conditions. When balancing a reaction under acidic conditions, it is imperative to balance the reaction using the following steps:
1.Divide each reaction into two ½ reactions
2.First balance elements other than H and O
3.Balance O by adding H2O as needed
4.Balance H by adding H+ as needed
5.Finally, balance chare by adding e- as needed
6.Multiply each half reaction so that there are equal number of electrons in each half reaction
7.Sum the reactions together (the electrons should cancel out).
1.Divide each reaction into two ½ reactions
2.First balance elements other than H and O
3.Balance O by adding H2O as needed
4.Balance H by adding H+ as needed
5.Finally, balance chare by adding e- as needed
6.Multiply each half reaction so that there are equal number of electrons in each half reaction
7.Sum the reactions together (the electrons should cancel out).
By adding hydrogen to each side, we create an acidic solution. —If the redox reaction occurs in basic solution, add equal amounts of OH- on each side after following the previously mentioned steps. The OH- reacts with the H+ to make water on one side of the equation and remains as OH- on the other side of the equation. Thus, our solution is now basic
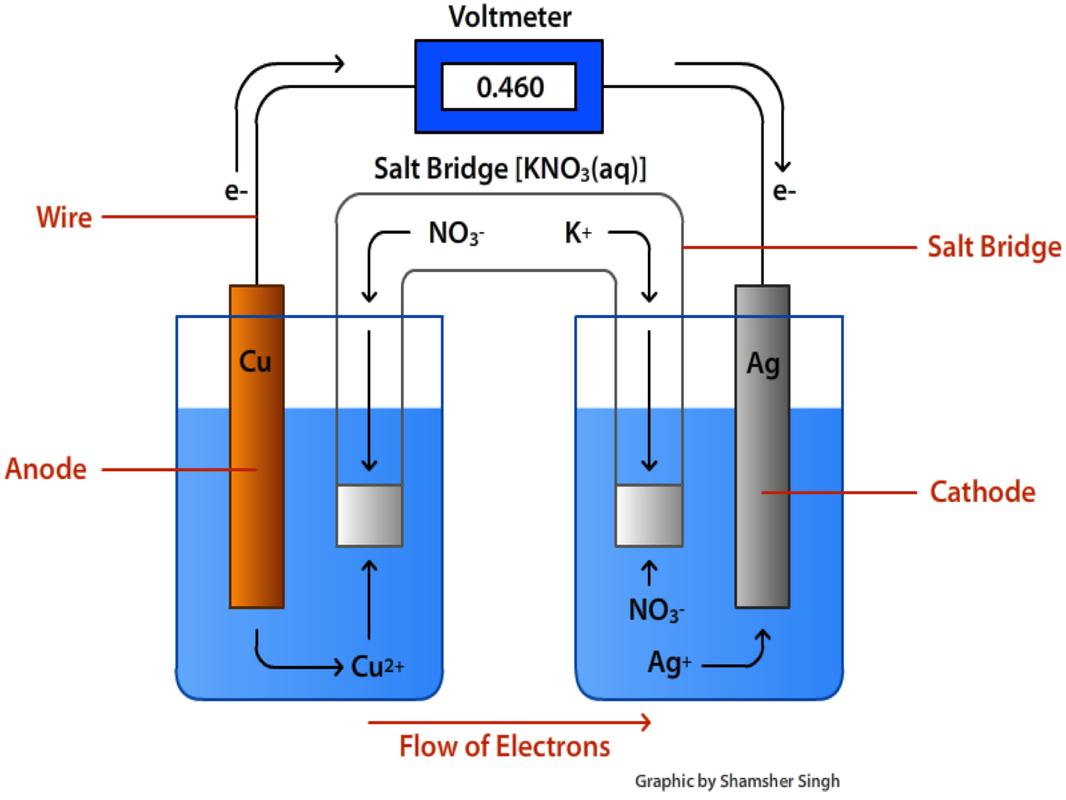
Galvonic (Voltaic) Cells
The energy released in a spontaneous redox reaction can be used to perform electrical work. —This is done through the construction of a voltaic cell. In a voltaic cell, the two half reactions are split and they are not in direct contact with one another. The movement of electrons between the two cells can be utilized to do work
The following components are crucial to the construction of a voltaic cell:
- Anode – The site where oxidation takes place. Negatively charged.
- Cathode – The site where reduction takes place. Positively charged.
- Salt Bridge – Connects the two ½ cells and contains an unreactive salt. This ensures that charge does not build up in the ½ cells. Common salts solutions are NaNO3 or KNO3
Standard Reduction Potentials
In a voltaic cell, electrons move from an area of high potential energy to an area of low potential energy. Differences in potential energy are measured in volts:
The potential difference between two cells is the cell potential (Ecell). When the cell is run under standard conditions, it is called the standard cell potential (Eocell).
We measure numerous half cells under standard conditions (25 oC, 1 M, and 1 atm). We can calculate the standard reduction potential (Eocell) for the electrochemical cell by utilizing:
We measure numerous half cells under standard conditions (25 oC, 1 M, and 1 atm). We can calculate the standard reduction potential (Eocell) for the electrochemical cell by utilizing:
Therefore, we can calculate the electrochemical potential for a voltaic cell simply by using a standrad reduction potential table like the one shown below:
The substance that causes another object to be oxidized is known as the oxidizing agent (it itself it reduced). —The substance that causes another object to be reduced is known as the reducing agent (it itself it oxidized). The higher you are on the reduction potential table, the better an oxidizing agent you are, The lower you are, the better a reducing agent you are when the reaction is reversed.
Nernst Equations
We can calculate standard reduction potentials using our reduction potential table, but we have a problem because most reactions do not occur under standard conditions (1 M, 25 oC, and 1 atm). Additionally, for a reaction, it proceeds until E = 0 (equilibrium has been achieved).
For those reactions run under non-standard conditions, we can use the Nernst equation:
For those reactions run under non-standard conditions, we can use the Nernst equation:
•E = Non-standard reduction potential
•Eo = Standard reduction potential
•R = Gas Constant = 8.314 J/(mol x K)
•T = Temperature (in Kelvin)
•n = Moles of electrons transferred
•F = Faraday’s constant = 96,500 C/mol
•Q = Reaction quotient (from balanced redox equation)
•Eo = Standard reduction potential
•R = Gas Constant = 8.314 J/(mol x K)
•T = Temperature (in Kelvin)
•n = Moles of electrons transferred
•F = Faraday’s constant = 96,500 C/mol
•Q = Reaction quotient (from balanced redox equation)
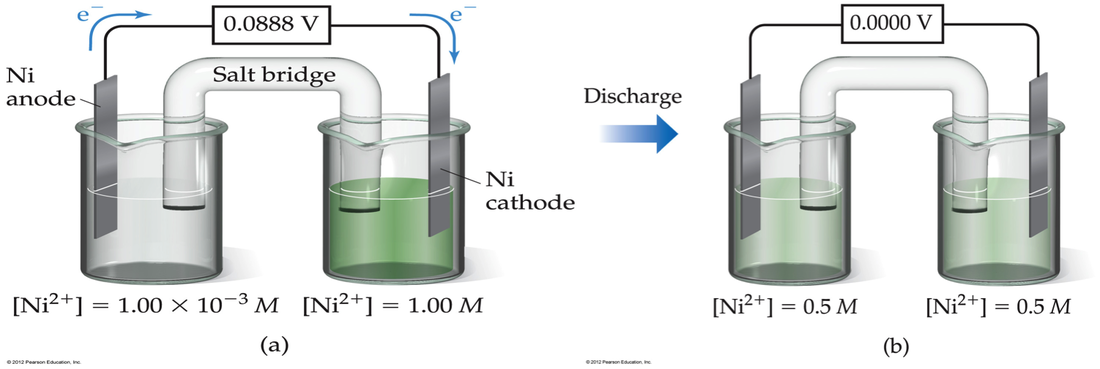
Concentration Cells
—In addition to difference in reduction potential, cell emf also depends on the concentration in each cell. Therefore, we can construct a cell where both the anode and cathode have the same ion, but there are different concentrations. The more dilute solution goes in the anode. The more concentrated solution goes in the cathode. The driving force is the entropy of mixing.
In the electrochemistry lab we studied the following concentration cell:
Therefore, the Eo for this solution would be:
If we add the reactions together we discover that the reaction is the concentrated going to the dilute Ag+:
Ag+ (concentrated) --> Ag+ (dilute)
—We can solve for E using the Nernst equation and Q is simply:
Free Energy, Equilibrium Constants, and E
Voltaic cells use a positive Eo in order to produce cell potential. Therefore, a Eo must be positive in order for a reaction to be spontaneous. This is why we can use standard reduction potentials to predict whether a metal will run in the forward or reverse reaction. Needs to generate a +Eo when reaction is ran.
Since the emf indicates whether a reaction is spontaneous we can use it to calculate ΔG:
Since the emf indicates whether a reaction is spontaneous we can use it to calculate ΔG:
Since n and F are always positive, the sign of E determines the spontaneity of a reaction!
Finally we can relate this to equilibrium constants:
Finally we can relate this to equilibrium constants:
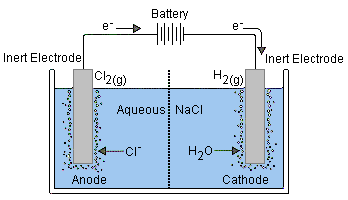
Electrolytic Cells
Recall, Eo is always a positive number for spontaneous chemical processes. We can cause a non-spontaneous process (-Eo value) to occur by attaching a battery and pumping electrons in the direction that they otherwise would not go in. The process of causing electrons to flow in the non-spontaneous direction with the assistance of a battery are known as electrolysis reactions or electrolytic cells.
These cells are different in that the two electrodes are immersed in either a molton salt or a solution der to facilitate the process (although the anode and cathode definitions do not change).
These cells are different in that the two electrodes are immersed in either a molton salt or a solution der to facilitate the process (although the anode and cathode definitions do not change).
The utility of electrolytic cells is that if we know the half reaction, the we can calculate the amount of substance reduced or oxidized in a given period of time just by knowing how many electrons have been passed in the cell.
The quantity of charge passing through an electrical circuit, such as that in an electrolytic cell, is generally measured in coulombs. As noted in before, the charge on 1 mole of electrons is 96,500 C. A coulomb is a quantity of charge passing a point in a circuit in 1 second when the current is 1 ampere (A). We can calculate coulombs by:
The quantity of charge passing through an electrical circuit, such as that in an electrolytic cell, is generally measured in coulombs. As noted in before, the charge on 1 mole of electrons is 96,500 C. A coulomb is a quantity of charge passing a point in a circuit in 1 second when the current is 1 ampere (A). We can calculate coulombs by:
Coulombs = Amperes x Seconds
Therefore, we can follow the following steps in order to convert to grams of a substance reduced or oxidized:
1. If the current and time are known, the coulombs can be calculated using the equation above
2. Coulombs can be converted to moles of electrons using Faraday's constant (96,500 C/mole of elections)
3. The balanced half reaction can be used to convert between moles of electrons and moles of the substance of interest
4. Once moles of the substance of interest is known, then you can convert to grams using the molar mass.
1. If the current and time are known, the coulombs can be calculated using the equation above
2. Coulombs can be converted to moles of electrons using Faraday's constant (96,500 C/mole of elections)
3. The balanced half reaction can be used to convert between moles of electrons and moles of the substance of interest
4. Once moles of the substance of interest is known, then you can convert to grams using the molar mass.
Practice, Practice, Practice!
|
Once you feel you have built up some endurance, try your hand at these practice problems. make sure to check your work once you are complete!
|
The Ultimate Showdown!
|
Do you feel you have built up enough endurance in order to go toe to toe with the best of the best? If yes, then take an attempt at this FRQ problem. NOTE - If you need to reattempt the FRQ, then please see Mr. Astor for a new problem.
|