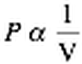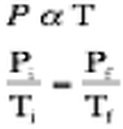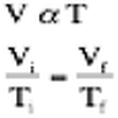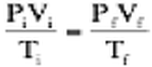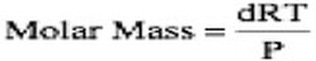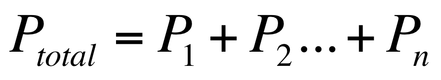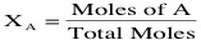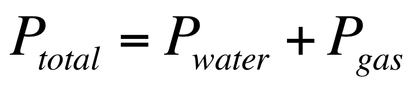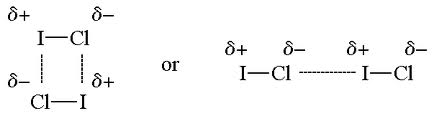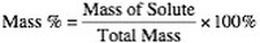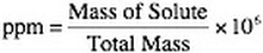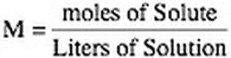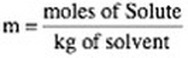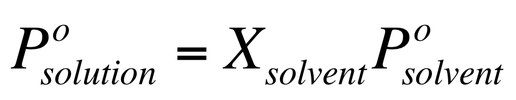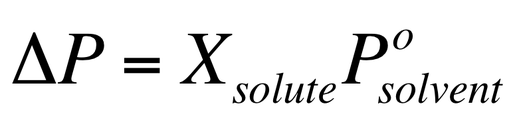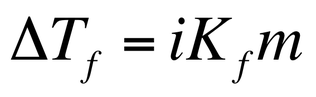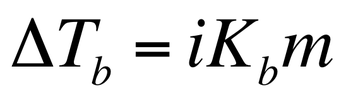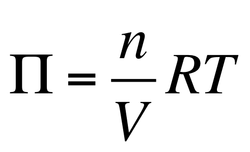|
No AP review would be complete without an ode to the Hunger Games. Channel your inner Katniss and go for the gold in this event that pushes you on your understanding of all the properties of solids, liquids and gases.
In order to hit the bullseye and receive a gold medal for your country, you will need to master the properties of solids, liquids, and gases. In addition, you will need to be able to perform the calculations for gases and solutions that were covered throughout the year. |
The Four Gas Laws
Last year we learned about three major gas laws that govern the behavior of all gases: Boyle’s, Charles’, and Gay-Lussac’s Law. These laws can be summarized as follows:
Boyle’s Law.
When we examine the pressure and volume of a gas, we notice that as the volume is decreased under constant temperature, then the pressure is increased. This is due to the fact that as the volume is decreased there is an increase in the amount of collisions of gas molecules with the surface of the container. This is what leads to the increase in pressure. This can be expressed by the function:
Boyle’s Law.
When we examine the pressure and volume of a gas, we notice that as the volume is decreased under constant temperature, then the pressure is increased. This is due to the fact that as the volume is decreased there is an increase in the amount of collisions of gas molecules with the surface of the container. This is what leads to the increase in pressure. This can be expressed by the function:
PiVi = PfVf
Gay – Lussac’s Law
The second gas law observed that as a gas was heated under constant volume, the pressure increased also. This is due to the fact that as the molecules are heated they gain more energy. This energy causes the gas molecules to move quicker and run into the walls of he container more frequently. This relationship can be described by the function:
The second gas law observed that as a gas was heated under constant volume, the pressure increased also. This is due to the fact that as the molecules are heated they gain more energy. This energy causes the gas molecules to move quicker and run into the walls of he container more frequently. This relationship can be described by the function:
Charles’ Law
The third, and final gas law that we studied last year, observed that as you heated a gas under constant pressure, the volume also increased. This was due to the fact that as the molecules are heated they gain more energy. This energy causes the gas molecules to move quicker and run into the walls of he container more frequently. This container was capable of expanding, so its volume increased proportionally. This can be described by the function:
The third, and final gas law that we studied last year, observed that as you heated a gas under constant pressure, the volume also increased. This was due to the fact that as the molecules are heated they gain more energy. This energy causes the gas molecules to move quicker and run into the walls of he container more frequently. This container was capable of expanding, so its volume increased proportionally. This can be described by the function:
Avogadro’s Law
There is a fourth law that is also included as a gas law, but we did not study it last year. This law is known as Avogadro’s Law and it relates moles to the overall volume of a container. This law states that as you increase the volume of a container, the moles of gas will also increase under constant temperature and pressure. This can also be described by the idea that 1 mole of any gas will occupy exactly 22.4 L.
Combined Gas Law
All of this culminates in the combined gas law, which literally combines the three gas laws that we studied last year. This law is represented by the expression:
There is a fourth law that is also included as a gas law, but we did not study it last year. This law is known as Avogadro’s Law and it relates moles to the overall volume of a container. This law states that as you increase the volume of a container, the moles of gas will also increase under constant temperature and pressure. This can also be described by the idea that 1 mole of any gas will occupy exactly 22.4 L.
Combined Gas Law
All of this culminates in the combined gas law, which literally combines the three gas laws that we studied last year. This law is represented by the expression:
The Ideal Gas Law
The ideal gas law is truly a remarkable equation. From this equation, we are able to relate the properties of a gas to the amount of moles. Once we know the amount of moles of a gas, then this equation can be manipulated in a wide variety of ways. The basic Ideal Gas Law states that:
PV = nRT
This equation has a few new variables, most notably, n and R. The variable n means moles of gas and R represents that gas constant (0.0821 (L x atm)/(mol x K)).
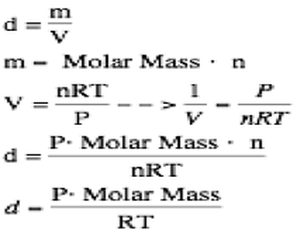
The true power of the ideal gas law comes from the fact that we can determine both the molar mass of an unknown gas and the density of this gas. We can determine the molar mass of the gas through the following idea:
PV = nRT
This equation has a few new variables, most notably, n and R. The variable n means moles of gas and R represents that gas constant (0.0821 (L x atm)/(mol x K)).
The true power of the ideal gas law comes from the fact that we can determine both the molar mass of an unknown gas and the density of this gas. We can determine the molar mass of the gas through the following idea:
You should not memorize this equation. You should be able to derive it because memorizing it will surely lead to you forgetting this equation before May! Finally, this equation can be used to calculate molar mass through a little rearrangement:
Dalton's Law of Partial Pressures
Dalton’s Law of Partial Pressure was developed based on the observation that the pressure of individual gas molecules was dependent only upon the volume of the container and not on the present of other gas molecules. This means that we can conclude that:
Dalton’s Law of Partial Pressure has two major implications:
1. We can characterize a gas by its mole fraction
2. We can collect a gas over water and determine the pressure of the gas so long as we cancel out the water vapor.
When examining the mole fraction of a compound, it is important to realize that we can determine the fraction of a certain gas mixture because the moles of each are not dependent on one another. Therefore, the mole fraction can be calculated by:
1. We can characterize a gas by its mole fraction
2. We can collect a gas over water and determine the pressure of the gas so long as we cancel out the water vapor.
When examining the mole fraction of a compound, it is important to realize that we can determine the fraction of a certain gas mixture because the moles of each are not dependent on one another. Therefore, the mole fraction can be calculated by:
This mole fraction can be multiplied by the total pressure in order to determine a partial pressure of a gas.
Furthermore, if we collect a gas over water, then we can calculate the pressure of the gas because it is known that:
Furthermore, if we collect a gas over water, then we can calculate the pressure of the gas because it is known that:
Kinetic Molecular Theory
|
Kinetic energy is the energy that is associated with any and all forms of motion. We can describe the motion of gas molecules in a similar way using kinetic molecular theory. There are 5 major postulates of KMT. These postulates state that:
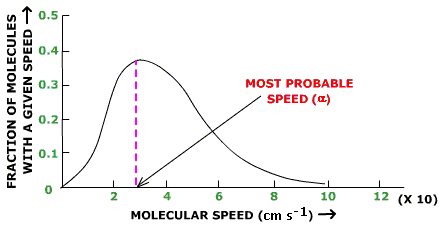
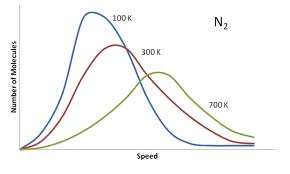
1. Gases consist of large numbers of molecule that are in continuous, random motion. 2. The size of gas molecules are negligible in comparison to the overall volume where the gas is contained. 3. Attractive and repulsive forces between gas molecules are negligible 4. Molecules inside a container collide, but the collisions result in no loss of energy and kinetic energy is conserved. 5. The average kinetic energy of the molecules is proportional to the temperature. Temperature provides a reading of the average kinetic energy of a sample of gas. These postulates allow for us to do the ideal gas law without having to consider attractive forces and/or the size of the gas molecules themselves. Furthermore, this theory provides a description about how the speeds of molecules are distributed. A hot gas has a wider distribution of molecular speeds than a cold gas whose distribution shows a higher frequency at a lower energy level. The same principle applies to light molecules vs. heavy molecules (heavy molecules move slower than light molecules). |
|
Intermolecular Forces
Intermolecular Forces are the forces that exist between molecules. These are much weaker than ionic, covalent, and metallic forces/bonds, but they still play a major role in chemical properties. The three types of intermolecular forces that we study are:
1. Hydrogen Bonding
2. Dipole – Dipole Forces
3. London Dispersion Forces
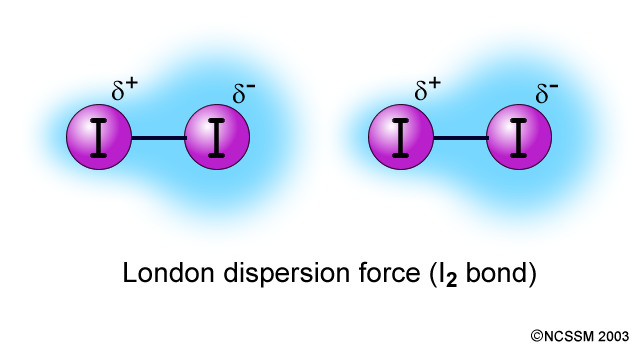
London dispersion forces are forces that exist in all atoms. In a neutral atom, electrons are spread evenly throughout a molecule. When two molecules are brought close enough to one another, electrons rearrange to create a temporary dipole. The polarizability of the molecule determines the strength of the dispersion force. Additionally, as molecules become heavier, the amount of dispersion forces also increases.
When a molecule has a permanent dipole, it has attractive forces to other dipoles Dipole – Dipole Forces are the attractive forces between molecules with partial positive and partial negative charges. As a molecule becomes heavier, the dipole – dipole force also becomes stronger.
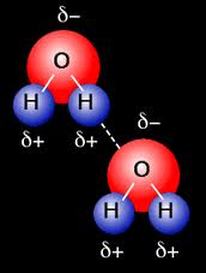
A special type of dipole – dipole force is known as hydrogen bonding. When H bonds to N, O, or F, there is a huge difference in electronegativity. Hydrogen bonding is the intermolecular force between the hydrogen bound to N, O, or F and another N, O, or F atom.
A few select properties are dependent upon the amount of intermolecular forces that a substance has. Namely, they are:
1. Freezing Point/Melting Point
2. Viscosity
3. Surface Tension
As the amount of intermolecular forces increases, the strength of each of these properties is also increased. This means that as you have stronger intermolecular forces, then the temperature at which a substance melts/freezes also is increased. Furthermore, more intermolecular forces leads to a higher surface tension and a more viscose material.
1. Hydrogen Bonding
2. Dipole – Dipole Forces
3. London Dispersion Forces
London dispersion forces are forces that exist in all atoms. In a neutral atom, electrons are spread evenly throughout a molecule. When two molecules are brought close enough to one another, electrons rearrange to create a temporary dipole. The polarizability of the molecule determines the strength of the dispersion force. Additionally, as molecules become heavier, the amount of dispersion forces also increases.
When a molecule has a permanent dipole, it has attractive forces to other dipoles Dipole – Dipole Forces are the attractive forces between molecules with partial positive and partial negative charges. As a molecule becomes heavier, the dipole – dipole force also becomes stronger.
A special type of dipole – dipole force is known as hydrogen bonding. When H bonds to N, O, or F, there is a huge difference in electronegativity. Hydrogen bonding is the intermolecular force between the hydrogen bound to N, O, or F and another N, O, or F atom.
A few select properties are dependent upon the amount of intermolecular forces that a substance has. Namely, they are:
1. Freezing Point/Melting Point
2. Viscosity
3. Surface Tension
As the amount of intermolecular forces increases, the strength of each of these properties is also increased. This means that as you have stronger intermolecular forces, then the temperature at which a substance melts/freezes also is increased. Furthermore, more intermolecular forces leads to a higher surface tension and a more viscose material.
Concentration Calculations
We recall that we create a solution by dissolving a solute in a solvent. Once this solution is created, there are numerous ways that we can calculate the concentration of that solution:
1. Mass Percent
1. Mass Percent
2. Parts per Million
3. Molarity
4. Molality
All of these equations are different ways of describing the ratio of solute to solvent/solution for a given mixture. All of these have their utility, but you need to be the one that decides how they are to be used based on the problem of interest.
Raoult's Law
The liquid state of a substance is in equilibrium with the gas state. The vapor pressure is the pressure when the solution is at this equilibrium. Solutions that have vapor pressure are said to be volatile.
We often dissolve a non-volatile substance in a volatile liquid. Raoult’s Law states that the partial pressure of a solvent vapor above a solution (Psolution) is the product of the mole fraction of the solvent (Xsolvent) and the pure vapor pressure (Psolvent):
We often dissolve a non-volatile substance in a volatile liquid. Raoult’s Law states that the partial pressure of a solvent vapor above a solution (Psolution) is the product of the mole fraction of the solvent (Xsolvent) and the pure vapor pressure (Psolvent):
We can also calculate the overall change in the vapor pressure when a solute is added by using the equation:
This change that is observed depends only on the amount of solute that is dissolved, not on the identity of the solute.
Colligative Properties
Colligative properties are properties that depend upon the amount of solute that is being dissolved, NOT the identity of the particular solute. There are four distinct examples of colligative properties. They are:
1. Vapor Point Lowering
2. Boiling – Point Elevation
3. Freezing Point Depression
4. Osmotic Pressure
When you dissolve a solute in a solvent, there are more solute particles that interact with the solvent, which prohibits the solvent from forming an ordered arrangement (freezing point) or breaking apart and leaving the solution (boiling point). This increased solute – solvent interactions create the freezing point lowering and boiling point elevation that is observed. The change in freezing point can be calculated by:
1. Vapor Point Lowering
2. Boiling – Point Elevation
3. Freezing Point Depression
4. Osmotic Pressure
When you dissolve a solute in a solvent, there are more solute particles that interact with the solvent, which prohibits the solvent from forming an ordered arrangement (freezing point) or breaking apart and leaving the solution (boiling point). This increased solute – solvent interactions create the freezing point lowering and boiling point elevation that is observed. The change in freezing point can be calculated by:
In this equation, i is the van’t hoff factor, Kf is the molar freezing point constant, and m is the molality of the solution. The change in the boiling point can be calculated by:
In addition to these two properties, we can calculate the osmotic pressure of a given solution. Osmosis is the movement of solvent through this membrane to the side of higher solute concentration. Osmotic Pressure (Π) is the pressure that will be required to stop osmosis from occurring:
All of these equations can be used to calculate the molar mass of given compounds. We can use any of the listed equations to solve for the number of moles (either through molality of molarity for the equations). If we know the mass of the compound dissolved, then we can use both of these values to calculate the molar mass.
Practice, Practice, Practice!
|
Now that you have covered the extensive amount of material that is necessary in order to hit the bulls eye in archery, complete the following practice problems in order to strengthen your overall skills.
|
The Main Event!
|
Make sure to have a steady arm and clear eye on the center of the target if you hope to achieve a medal in this event. NOTE - if you need to retake the FRQ, please see Mr. Astor for a new FRQ
|