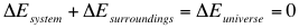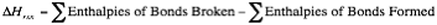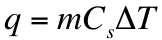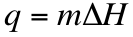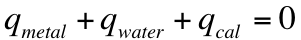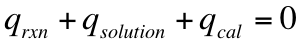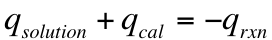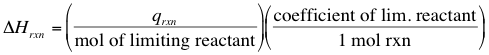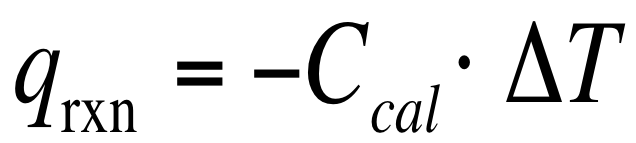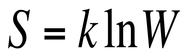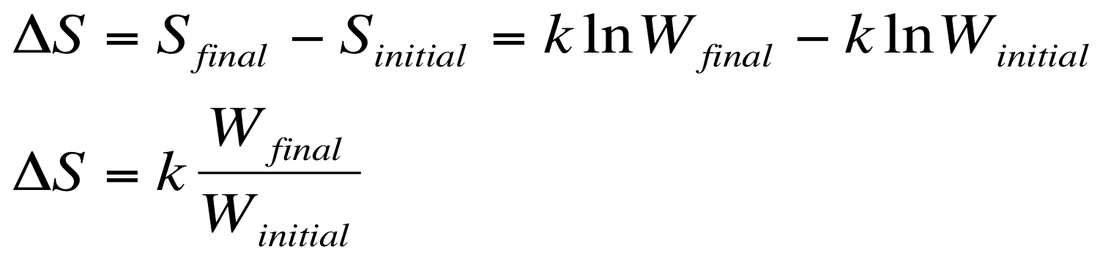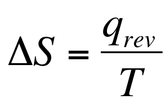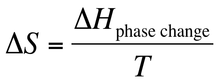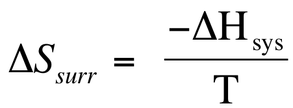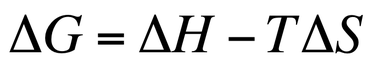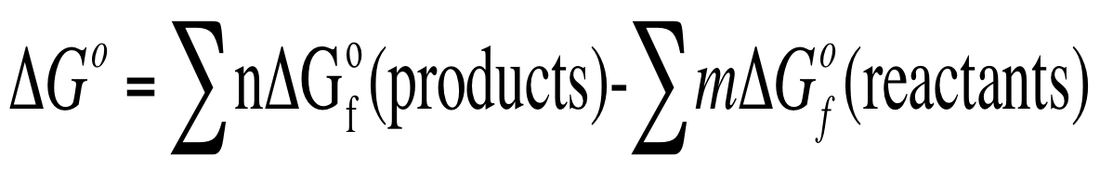Soccer - Thermodynamics
|
One of the most competitive sports across the world is soccer. Both the Latin America and European countries have long dominated this sport. Now, even North America and Asia are beginning to be competitive in a sport that they used to lose at frequently.
In order to be standing with the gold medal at the end of the championship game, you are going to need to be the master of all things thermodynamics. This includes enthalpy (∆H), entropy (∆S), and (∆G). If you can manipulate these values correctly, then you are sure to be the champion on the soccer field. |
First Law of Thermodynamics, Heat, Work, and Enthalpy
When examining chemical reactions, we can begin to describe the energetics of these reactions. This description allows for reactions to be characterized based on the amount of energy they release or absorb. As previously mentioned energy can be either kinetic or potential. Energy can come in one of two forms: work and heat. When looking at reactions, it is important to recognize that we need to examine the total amount of energy change. We use ∆E (or change in internal energy) to describe the total amount of energy:
∆E = q + w
When looking at this equation, we can begin to study the First Law of Thermodynamics. The 1st law simply states that energy cannot be created nor destroyed. It can only change forms. This means that if our system loses energy, then all of this energy must be lost to the surroundings. This is described as:
∆E = q + w
When looking at this equation, we can begin to study the First Law of Thermodynamics. The 1st law simply states that energy cannot be created nor destroyed. It can only change forms. This means that if our system loses energy, then all of this energy must be lost to the surroundings. This is described as:
∆E is an example of a state function. It doesn’t matter the path that the substance took to get to that state, the only measurements that mater are the initial and final conditions.
The final value that we examined when looking at the energetics of a reaction is the change in enthalpy (∆H). Enthalpy is a measurement of the total energy that goes in and out of the system. When it is measured under constant pressure, we can conclude that:
The final value that we examined when looking at the energetics of a reaction is the change in enthalpy (∆H). Enthalpy is a measurement of the total energy that goes in and out of the system. When it is measured under constant pressure, we can conclude that:
∆H = q (under constant pressure)
|
We characterize numerous reactions based on the ∆H that they produce. Recall, a +∆H is an endothermic process and a -∆H is an exothermic process. We can characterize numerous processes as being endothermic or exothermic based solely upon their enthalpy values.
The first type of enthalpy that we describe is the enthalpy of a reaction. This is the change in enthalpy that is associated with a chemical reaction. When we say enthalpy of reaction, what we are basically saying is: |
What this equation says is that if you only examine the energy needed to break apart all the individual bonds in a reaction and then the energy required to make all of the new bonds, then the enthalpy of the reaction can be calculated.
When a substance undergoes a phase change, there is heat that is required in order for this to occur. Turning a solid to a liquid to a gas required energy, so these are endothermic processes. Turning a gas to a liquid to a solid releases energy, so these are exothermic processes. The energy associated with these processes can be represented with ∆H values. Remember, the enthalpy of a solid to a liquid is the same value as a liquid to a solid, but the two have opposite signs.
When a substance undergoes a phase change, there is heat that is required in order for this to occur. Turning a solid to a liquid to a gas required energy, so these are endothermic processes. Turning a gas to a liquid to a solid releases energy, so these are exothermic processes. The energy associated with these processes can be represented with ∆H values. Remember, the enthalpy of a solid to a liquid is the same value as a liquid to a solid, but the two have opposite signs.
Using these enthalpy values, we can use Hess’s Law to calculate the ∆H for a given reaction. Hess’s Law states that if we know the steps of a reaction, then we can add the ∆H for these reactions together in order to determine the ∆H for the reaction of interest. There are two major points that need to be remembered when using Hess’s Law:
1. If a reaction is run in reverse, then the sign of ∆H is reversed.
2. If you multiple a reaction by a coefficient, then ∆H is multiplied by that same coefficient.
The final type of enthalpy value that we focused on in this unit are enthalpies of formation. These are enthalpies that are associated when we take elements in their standard state and react them to form the compound of interest. This means that if we are interested in how CO2 is formed, then we would describe its enthalpy of formation by the equation:
1. If a reaction is run in reverse, then the sign of ∆H is reversed.
2. If you multiple a reaction by a coefficient, then ∆H is multiplied by that same coefficient.
The final type of enthalpy value that we focused on in this unit are enthalpies of formation. These are enthalpies that are associated when we take elements in their standard state and react them to form the compound of interest. This means that if we are interested in how CO2 is formed, then we would describe its enthalpy of formation by the equation:
Elements always have enthalpies of formation that are zero. In addition to being very useful when characterizing a certain subset of reaction, ∆H of formation are useful because they provide a shortcut for calculating ∆H for a reaction. If the ∆H of formation values are known for all of the compounds in a reaction, then the ∆H for a reaction can be determined through the equation:
C (s) + H2 (g) --> CO2 (g)
Calorimetry
|
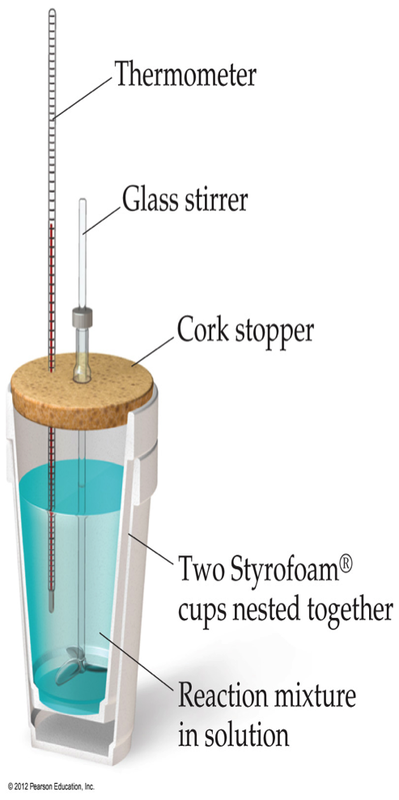
Now that we understand the theoretical idea of ∆H and how it can be calculated from both q and other ∆H values. It is important to begin to discuss how we can go into the lab and measure ∆H for a given chemical reaction. Calorimetry provides the tool by which scientists can sole these problems.
We can go into the lab and measure the heat that is generated in a reaction. This heat that is produced can be calculated in one of two ways. If there is a temperature change than q can be calculated as: If the substance is undergoing a phase change, then the heat produced can be calculated by:
It is important to note that ∆H can have units of either J/mol or J/g. Depending on the unit, you must determine whether you multiply by grams or moles of substance.
These two equations allow for us to characterize numerous aspects of a chemical reaction/process. It is important to note though that the total heat of a process is merely the sum of all the individual q terms for a process. This means that if we take a hot metal and place it in liquid water and no heat is lost to the surroundings (i.e. it is placed in a calorimeter), then the sum of the q terms looks like: We can do this because of the 1st Law of Thermodynamics! If we close our system off from the outside world inside a calorimeter, then the sum of the q terms must be equal to 0. To start these problems, you need to identify the number of q terms. Remember, if you are undergoing multiple phase changes, then each of these gets a separate q term.
|
One of the biggest uses for calorimetry is it allows for us to measure the heat that is produced during a chemical reaction. We know that any heat that is produced by a reaction must be absorbed/taken from the solution and the calorimeter. This means that we can say that:
We can not directly measure the heat produced in a reaction (q rxn), but we can measure how much heat is absorbed by the solution and the surroundings. Therefore, we can conclude that:
From the heat of reaction, we can calculate the ∆H for the reaction! So long as we identify the limiting reactant for the reaction that is studied, we can use the following equation:
|
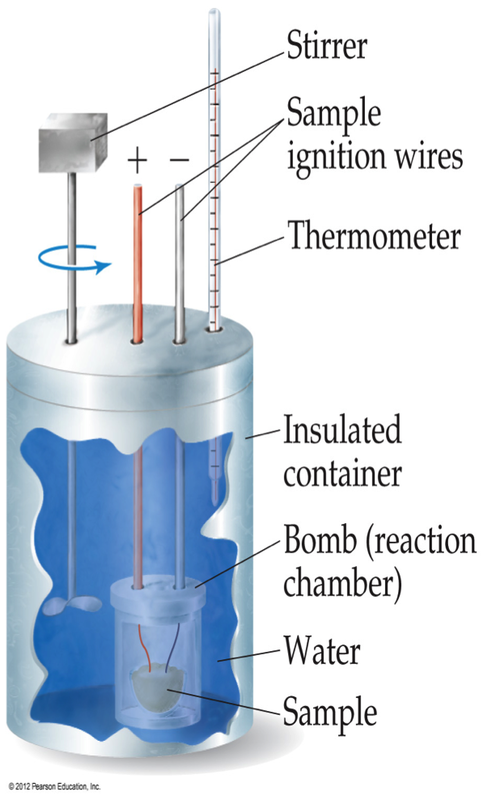
There is one other type of calorimetry and that is constant volume calorimetry. Keep in mind that the coffee cup calorimeter is designed so as to ensure that pressure can remain constant. This is extremely useful when studying the thermodynamics of a combustion reaction. Under constant volume calorimetry, you only need to know the change in temperature and the calorimeter constant and then you can use the equation:
Keep in mind, this heat of reaction is calculated under constant volume, not constant pressure. This means, we can only calculate ∆E from these heats of reactions. We cannot calculate ∆H.
|
Entropy and Microstates
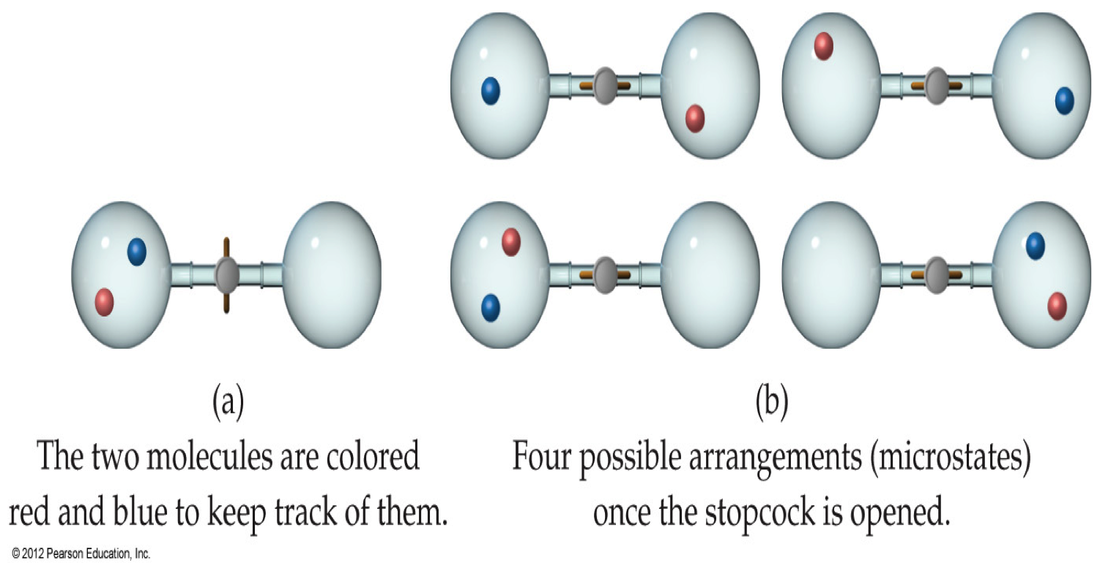
Why do gases choose to spread out? When we release a gas, past experience tells us that this gas will spread out when we spray it from a can. But we, as scientists, need an explanation as to why this process is observed. Imagine a gas with two molecules. If these are in a container, then there are 4 possible arrangements that these gas molecules:
We can see that there is a 50% chance that we will have one gas molecule on the left and that we have only a 25% chance of getting all the gas molecules on the left. As we increase the number of gas molecules in this system, the probability of all the gas molecules remaining on the left side becomes lower and lower unit the statistical possibility approaches zero.
This tendency towards disorder in a given system is known as entropy. This observation is a very commonly observed one in our daily lives, but we can calculate the entropy at a given point in time using the equation:
This tendency towards disorder in a given system is known as entropy. This observation is a very commonly observed one in our daily lives, but we can calculate the entropy at a given point in time using the equation:
In the above equation, S represents entropy, k is Boltzmann's constant, and W is the number of microstates. A microstate is one possible arrangement of the atoms in a system. Therefore we can see that the more microstates a system has, the higher the entropy of the system.
We care more about the change in entropy of a system. So the ∆S can be calculated by:
We care more about the change in entropy of a system. So the ∆S can be calculated by:
Therefore, we can see that an increase in microstates leads to a +∆S and a decrease in microstates leads to a -∆S value. When examining processes, we can make the following qualitative predictions about reactions:
- An increase in volume, leads to an increase in ∆S
- An increase in temperature, leads to an increase in ∆S
- An increase in the number of molecules from reactants to products, leads to an increase in ∆S
The 2nd and 3rd Law of Thermodynamics
Now that we know how to describe a reaction using both enthalpy (∆H) and entropy (∆S), we can begin discussing the final two laws of thermodynamics. The 1st law is great, but it does not tell us whether a reaction will occur or not. Therefore, we need new laws to describe this.
When describing chemical reactions, a process/reaction that will proceed without outside assistance is known as a spontaneous process. A process that requires work in order to proceed is known as a non-spontaneous process. We need to be able to describe these processes.
When describing chemical reactions, a process/reaction that will proceed without outside assistance is known as a spontaneous process. A process that requires work in order to proceed is known as a non-spontaneous process. We need to be able to describe these processes.
|
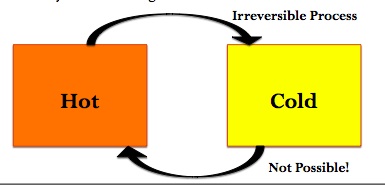
Recall, energy can come in two forms: heat and work. Let us examine these two forms. If you have a hot object near a cold object, then the heat will flow from hot to cold, but never from cold to hot. This means that the transfer of heat is an irreversible process.
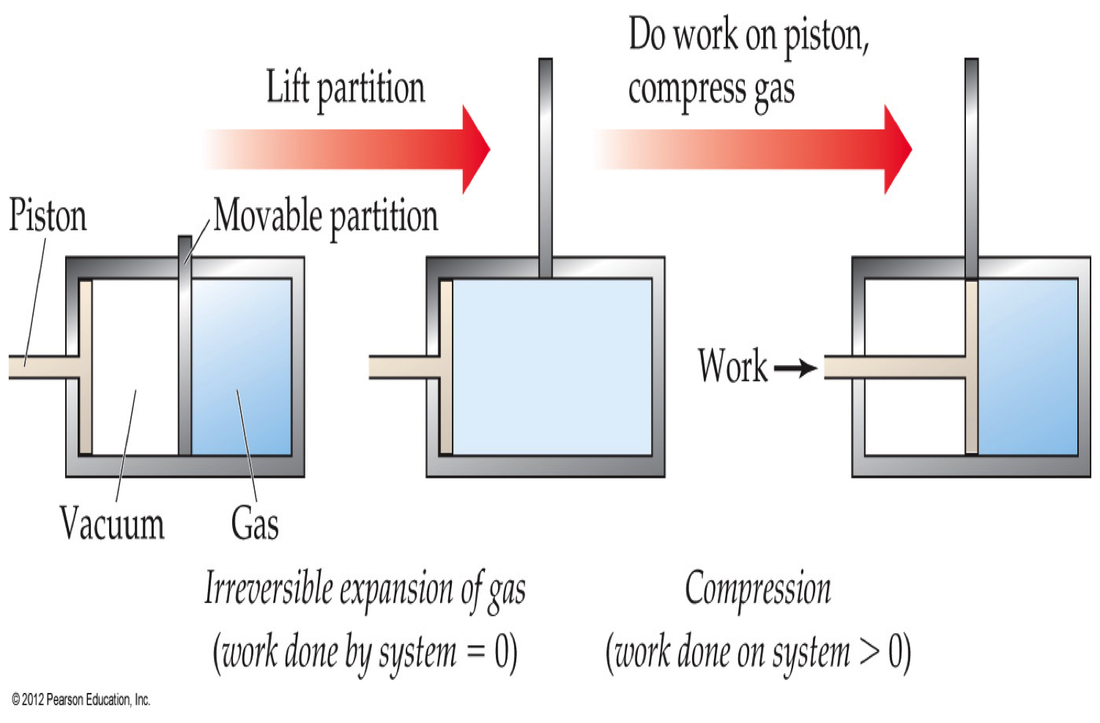
To examine work, consider an isothermal process (any process run under constant temperature). If we have a gas that is divided so that half of its container is a vacuum and the other half contains the gas. If we remove the divider, then gas will expand spontaneously without the need for work (due to entropy). If we then push down on a piston, then work is exerted on the gas in order to return it to the initial volume. Therefore, we needed to put work in for a process that initially required no work. This allows us to conclude that work is an irreversible process. This leads to the dramatic conclusion that:
|
We can then use this to calculate ∆S because if a spontaneous process is irreversible, the heat generated in the process is a useful tool by which to describe the reaction. We can calculate ∆S by saying that:
We are able to now calculate the ∆S of a the surroundings and system for a given process. When we calculate ∆S we see that the 2nd Law of Thermodynamics is:
This allows for us to begin to characterize a wide variety of chemical reactions, because we now can interconvert between enthalpy and entropy. We know that as we increase entropy of the surroundings, then we know that this was increased due to a loss of enthalpy from the system (-∆H). We also know that the entropy of the surroundings will increase as temperature is increased, so we can say that:
We now have numerous equations to characterize ∆S. We need to ask ourselves the question though of when entropy is zero. If we look at the Boltzmann equation, we can see that S = k ln W. We therefore can see that the only point where we will get S = 0 is when W is 1.
The only point where entropy can be zero is when we have 1 microstate possible. The only way to achieve one microstate is ensuring that molecules cannot move. Therefore, the 3rd law of thermodynamics states that:
The only point where entropy can be zero is when we have 1 microstate possible. The only way to achieve one microstate is ensuring that molecules cannot move. Therefore, the 3rd law of thermodynamics states that:
3rd Law: Entropy of a pure crystalline substance at absolute zero is 0.
Entropy can only cease when we have reached the point of absolute zero!
Standard Entropies (So)
We use calorimetry to measure ΔH, but we do not have a nice way to measure ΔS in a reaction. Due to the 3rd Law, we know that every substance must have a point where S = 0. As temperature increases from 0 K, the value of S also increases. We therefore can determine standard entropies (So).
Standard Entropies is the entropy gained by taking 1 mol of perfect crystalline substance at 0 K and bringing it to standard conditions. It is measured in J/(mol x K). All elements and compounds have standard entropies and the value of these increase as the weight of a substance increases.
Using these known standard entropies, we can calculate ΔS for reactions using the equation:
Standard Entropies is the entropy gained by taking 1 mol of perfect crystalline substance at 0 K and bringing it to standard conditions. It is measured in J/(mol x K). All elements and compounds have standard entropies and the value of these increase as the weight of a substance increases.
Using these known standard entropies, we can calculate ΔS for reactions using the equation:
Predicting Spontaneity (ΔG)
Recall, in chemistry, a spontaneous reaction/process is one that will occur without any additional assistance (electricity, new chemicals, etc.). Reactions are spontaneous based on their values of of ΔH and ΔS. We determine if a reaction is spontaneous or not based on the value of Gibbs Free Energy (ΔG). ΔG can be calculated by utilizing the following equation:
In the above equation, ΔG is measured in kJ/mol, ΔH is measured in kJ/mol, T is measured in Kelvin, and ΔS is measured in J/(mol x K). Make sure your units agree when doing these calculations!
Using Gibbs Free Energy alone we will be able to predict the spontaneity of a reaction. The value of ΔG indicates one of three options:
1. If ΔG < 0, then reaction is spontaneous as written
2. If ΔG = 0, then reaction is at equilibrium
3. If ΔG > 0, then reaction is not spontaneous as written (spontaneous in reverse direction)
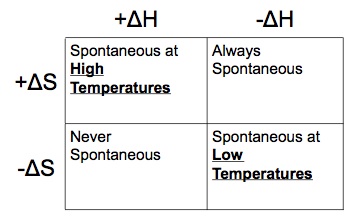
Therefore, we can examine how positive or negative values of ΔH and ΔS encourage or discourage spontaneous chemical reactions by using the table below. NOTE - If you are asked to calculate than it is spontaneous at that specific temperature. This table is used only to make qualitative predictions.
Using Gibbs Free Energy alone we will be able to predict the spontaneity of a reaction. The value of ΔG indicates one of three options:
1. If ΔG < 0, then reaction is spontaneous as written
2. If ΔG = 0, then reaction is at equilibrium
3. If ΔG > 0, then reaction is not spontaneous as written (spontaneous in reverse direction)
Therefore, we can examine how positive or negative values of ΔH and ΔS encourage or discourage spontaneous chemical reactions by using the table below. NOTE - If you are asked to calculate than it is spontaneous at that specific temperature. This table is used only to make qualitative predictions.
Standard Free Energies of Formation (ΔGo)
The standard free energy of formation (ΔGfo) is the change in free energy when we take elements in their standard state to make a compound. Same idea as ΔHfo. We can use these to calculate ΔGo even when ΔSo and ΔHo are unknown.
Practice, Practice, Practice!
|
Once you feel you have adequately completed the material that is covered on this site, try your hand at these drills so that you can be ready for the championship game!
|
The Championship Game!
|
When you feel that you have achieved an understanding that will allow for you to take down the best soccer players in the country, then you can complete the FRQ problem for this section. NOTE - If you need to retake this FRQ, then you need to see Mr. Astor for additional FRQ problems.
|