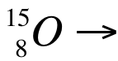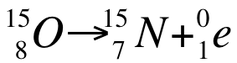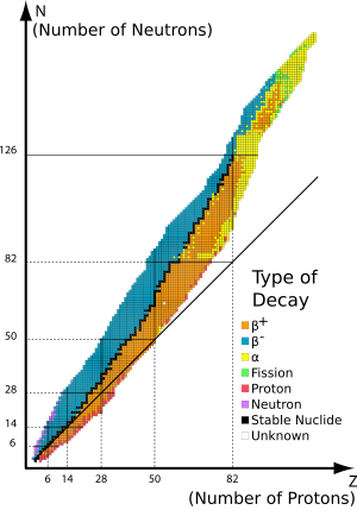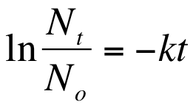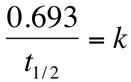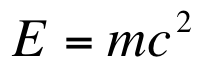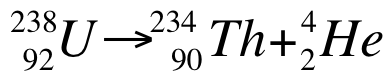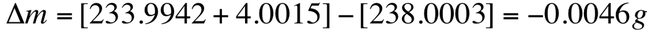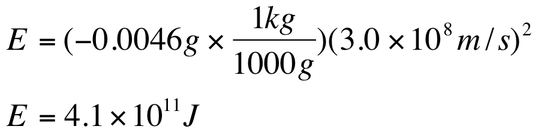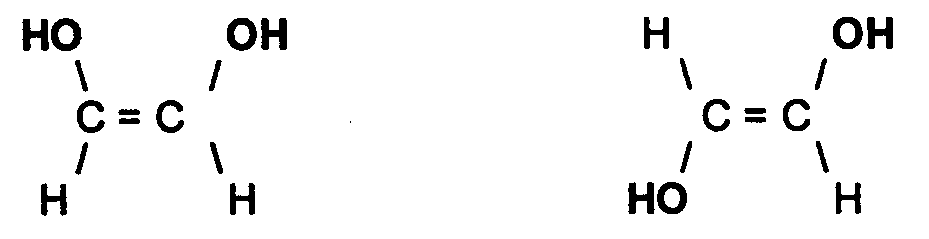Beach Volleyball - Nuclear and Organic Chemistry
|
A sport that is near and dear to Southern California residents is beach volleyball. What can be more fun than being able to dive for every ball because you have sand to break your fall? This sport has been long dominated by countries in the warmer climates (for some reason Russian beaches are hard to come by...), but there are always bound to be a few surprises.
In order to emerge victorious from this event you will need to have a deep understanding of nuclear equations, nuclear decay, half lives, Einstein's equation for energy, and a general understanding of introductory organic chemistry. |
Nuclear Decay/Equations
Recall, atoms of the same element can have different masses. Atoms that are the same mass, but have a different mass (due to a difference in the number of neutrons in the nucleus) are known as isotopes. Some isotopes are stable, but many of these isotopes breakdown over time. Based on the number of protons and neutrons in the nucleus, an element may be radioactive and spontaneously breakdown overtime. During this breakdown particles and/or emissions are released from the nucleus of the atom of interest.
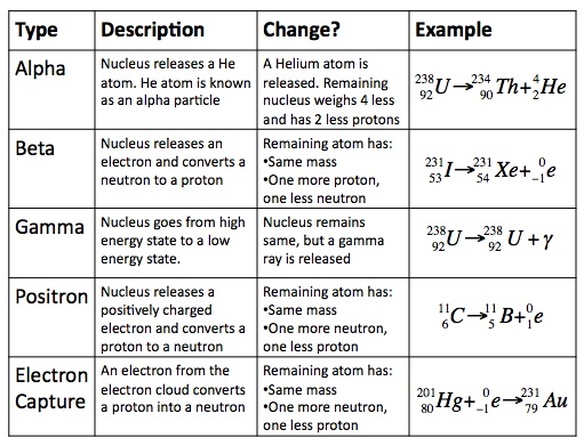
There are five main types of decay that a molecule can undergo:
1. Alpha Decay
2. Beta Decay
3. Gamma Decay
4. Positron Emission
5. Electron Capture
Atoms undergo decay due to a wide array of reasons, but the majority of atoms undergo these five types of characteristic decays. Each type of decay is unique, but you need to be able to understand what is happening in each type of decay in order to be successful in competition. The table below summarizes each type of decay:
There are five main types of decay that a molecule can undergo:
1. Alpha Decay
2. Beta Decay
3. Gamma Decay
4. Positron Emission
5. Electron Capture
Atoms undergo decay due to a wide array of reasons, but the majority of atoms undergo these five types of characteristic decays. Each type of decay is unique, but you need to be able to understand what is happening in each type of decay in order to be successful in competition. The table below summarizes each type of decay:
In addition to being able to identify a reaction as one of the five types of decay, you need to be able to write nuclear equations (like we did last year). In order to achieve this, you need to start by writing out the reactants given in the problem. For example, if asked to write the nuclear equation for 15 - oxygen undergoing positron emission, start by recognizing that we need to write out the formula for 15 - oxygen, which is:
Next we know that positron emission requires the release of a positively charged electron and a proton is converted to a neutron. This means the mass number will remain at 15, but the atomic number will decrease to 7. This means we are no longer O, rather we are now a nitrogen atom. Therefore, the final nuclear equation can be written as:
Patters of Nuclear Stability
The stability of the nucleus is directly dependent upon the number of neutrons and protons that are contained within the nucleus of the atom. Recall, inside the nucleus of our atom there is a strong nuclear force. This is the force between protons and neutrons and it is what prevents our nucleus from splitting apart due to the positive-positie repulsion between protons. As the number of protons increases inside the nucleus, there is a greater need for neutrons to counteract the proton-proton repulsion. Therefore, the proton to neutron ratio is critical to the stability of an atom and whether the atom is radioactive or not.
The graph below illustrates the type of stable nuclei in relation to the ratio of protons to neutrons. You can see that as the number of protons increase, an even larger ratio of neutrons is required inside the atom in order to maintain stability. The black jagged line represents our "belt of stability":
The graph below illustrates the type of stable nuclei in relation to the ratio of protons to neutrons. You can see that as the number of protons increase, an even larger ratio of neutrons is required inside the atom in order to maintain stability. The black jagged line represents our "belt of stability":
The type of radioactive decay that a particular radioactive atom exhibits is largely dependent upon the neutron-to-proton ratio. There are 3 general properties we can take from this graph
1. Nuclei above the belt of stability - These are neutron rich atoms need more protons, so they undergo beta decay to convert a neutron to a proton.
2. Nuclei below the belt of stability - These are proton rich atoms that need more neutrons, therefore they undergo either electron capture or positron emission.
3. Nuclei with atomic number above 84 - All atoms above 84 are radioactive. These heavy nuclei typicall undergo alpha decay to become smaller.
1. Nuclei above the belt of stability - These are neutron rich atoms need more protons, so they undergo beta decay to convert a neutron to a proton.
2. Nuclei below the belt of stability - These are proton rich atoms that need more neutrons, therefore they undergo either electron capture or positron emission.
3. Nuclei with atomic number above 84 - All atoms above 84 are radioactive. These heavy nuclei typicall undergo alpha decay to become smaller.
Half Lives
While many different types of nuclei undergo radioactive decay, different nuclei undergo radioactive decay at different rates. Nuclei can decay in a mater of seconds or it can take millions (or billions!) of years. Therefore, we can begin to describe the rate of nuclear decay.
All radioactive decay is a first - order kinetic process. This means that we can use the same first order integrated rate law equations that we utilized in the kinetics unit. Recall, if we know the initial amount of a substance and the amount of time that has elapsed, then we can use the following integrated rate law:
All radioactive decay is a first - order kinetic process. This means that we can use the same first order integrated rate law equations that we utilized in the kinetics unit. Recall, if we know the initial amount of a substance and the amount of time that has elapsed, then we can use the following integrated rate law:
Where No represents the amount of atoms initially, Nt represents the amount of atoms at time t, t represents the time that has elapsed, and k is the rate constant. Therefore, the amount of a sample that remains after a given amount of time can be calculated using only this equation!
A very common measurement in nuclear chemistry is half-lives. The half-life of a substance is the amount of time it takes until the substance is half what it was initially. For example, if a 5 gram sample has a half life of 2 years, then this means that after 2 years there will be 2.5 g of the original substance remaining. After 4 years, it will have undergone another half-life and there will now be 1.25 g remaining. This process will continue until nearly all of the substance has decayed. Using the above equation, we discover that the half-life can be used with the equation:
A very common measurement in nuclear chemistry is half-lives. The half-life of a substance is the amount of time it takes until the substance is half what it was initially. For example, if a 5 gram sample has a half life of 2 years, then this means that after 2 years there will be 2.5 g of the original substance remaining. After 4 years, it will have undergone another half-life and there will now be 1.25 g remaining. This process will continue until nearly all of the substance has decayed. Using the above equation, we discover that the half-life can be used with the equation:
Einstein's Equation
We recall that Einstein's famous equation is:
This equation explains that mass and energy are directly proportional. If we lose any mass, then it must converted into energy (a very large amount since we are multiplying by the speed of light squared!).
In nuclear reactions, there is a noticeable change in mass between reactants and products. The mass changes lead to a release of a large amount of energy. For example, the following nuclear decay:
In nuclear reactions, there is a noticeable change in mass between reactants and products. The mass changes lead to a release of a large amount of energy. For example, the following nuclear decay:
The masses of the nuclei are uranium = 238.0003 amu, Th = 233.9942 amu, and He = 4.0015 amu. The mass change is the mass of the products subtracted from the mass of the reactants:
This change in mass must have been converted into energy, so we can calculate this energy with Einstein's equation:
FInally, we have known that the masses of nuclei are always less than the masses of the individual subatomic particles that make up the atom. This difference between the mass of the nucleus and the mass of the subatomic particles is called the mass defect.
The energy required to separate a nucleus into its individual nucleons is called the nuclear binding energy and we can utilize Einstein's equation due to the fact that the mass defect is our "m" term. If m is known, then we can calculate the energy that is required to break apart this atom into its subatomic particles.
The energy required to separate a nucleus into its individual nucleons is called the nuclear binding energy and we can utilize Einstein's equation due to the fact that the mass defect is our "m" term. If m is known, then we can calculate the energy that is required to break apart this atom into its subatomic particles.
Hydrocarbons (Alkanes, Alkenes, and Alkynes)
Organic chemistry is the study of carbon. Because of carbons ability to bond with a wide variety of substances, carbon forms the backbone of most molecules that we find on this planet. Carbon can form a backbone that can be substituted with molecules such as O, H, and N in order to create a vast diversity of compounds.
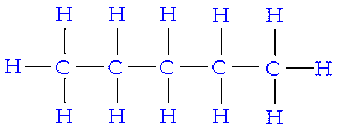
Because carbon compounds are so numerous, it is convenient to organize them into families that have structural similarities The simplest class of organic compounds are hydrocarbons (compounds with only hydrogen and carbon). Hydrocarbons have a chain of carbon-carbon bonds with the other empty spaces being filled by hydrogen. An example of a 5 carbon hydrocarbon can be seen below:
Because carbon compounds are so numerous, it is convenient to organize them into families that have structural similarities The simplest class of organic compounds are hydrocarbons (compounds with only hydrogen and carbon). Hydrocarbons have a chain of carbon-carbon bonds with the other empty spaces being filled by hydrogen. An example of a 5 carbon hydrocarbon can be seen below:
Hydrocarbons can be broken down into 4 main categories:
1. Alkanes - Contain only single bonds
2. Alkenes - Contain at least one double bond
3. Alkynes - Contain at least one triple bond
4. Aromatic Hydrocarbons - Carbons bound in a ring structure with delocalized pi bonds above and below the ring.
The boiling point of hydrocarbons are characterized by two factors: length of hydrocarbon and number of double/triple bonds. The longer the chain, the higher the boiling point becomes due to the fact that the molecule has a greater overall surface area. Furthermore, as you add more double and triple bonds, the boiling point increases due to the increased amount of surface area of the compound.
1. Alkanes - Contain only single bonds
2. Alkenes - Contain at least one double bond
3. Alkynes - Contain at least one triple bond
4. Aromatic Hydrocarbons - Carbons bound in a ring structure with delocalized pi bonds above and below the ring.
The boiling point of hydrocarbons are characterized by two factors: length of hydrocarbon and number of double/triple bonds. The longer the chain, the higher the boiling point becomes due to the fact that the molecule has a greater overall surface area. Furthermore, as you add more double and triple bonds, the boiling point increases due to the increased amount of surface area of the compound.
Isomers
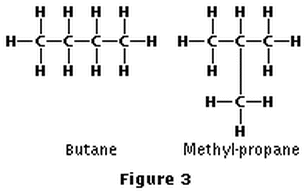
Oftentimes, we have molecules that have the same molecular formula, but there are multiple ways in which the molecule can be arranged in space. Compounds that have the same molecular formula, but a different bonding arrangement (two or more different structures when drawn) are known as isomers. The two pictures below represent two possible isomers of butane (C4H10). This is known as structural isomers:
As the number of carbons in the alkane increases, so to do the number of possible isomers that the molecules can take.
Furthermore, there are also geometric isomers whereby, we have compounds that have the same molecular formula and the same groups bonded to one another, but differ in the spatial arrangement of these groups. These are known as geometric isomers. An example of a geometric isomer can be seen below:
Furthermore, there are also geometric isomers whereby, we have compounds that have the same molecular formula and the same groups bonded to one another, but differ in the spatial arrangement of these groups. These are known as geometric isomers. An example of a geometric isomer can be seen below:
Naming Alkanes, Alkenes, and Alkynes
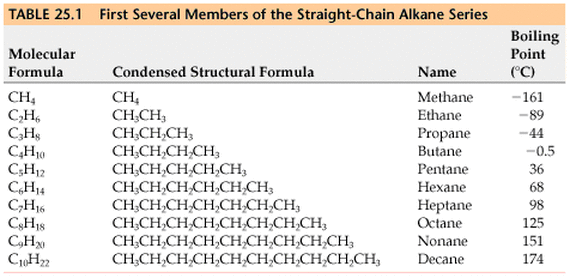
We have a systematic way by which we can name alkanes. Based on the number of carbons on the longest hydrocarbon chain, we attach a prefix that ends with the root -ane in order to signify the number of carbons. The roots for 1 - 10 carbon long chains are seen in the table below:
If the longest hydrocarbon chain contains a double bond, then the -ane suffix is replaced by -ene (i.e. propene, butene, pentene, etc). If the longest hydrocarbon chain contains a triple bond, then the -ane suffix is replaced with -yne (i.e. butyne, pentyne, hexyne, etc.).
Organic Functional Groups
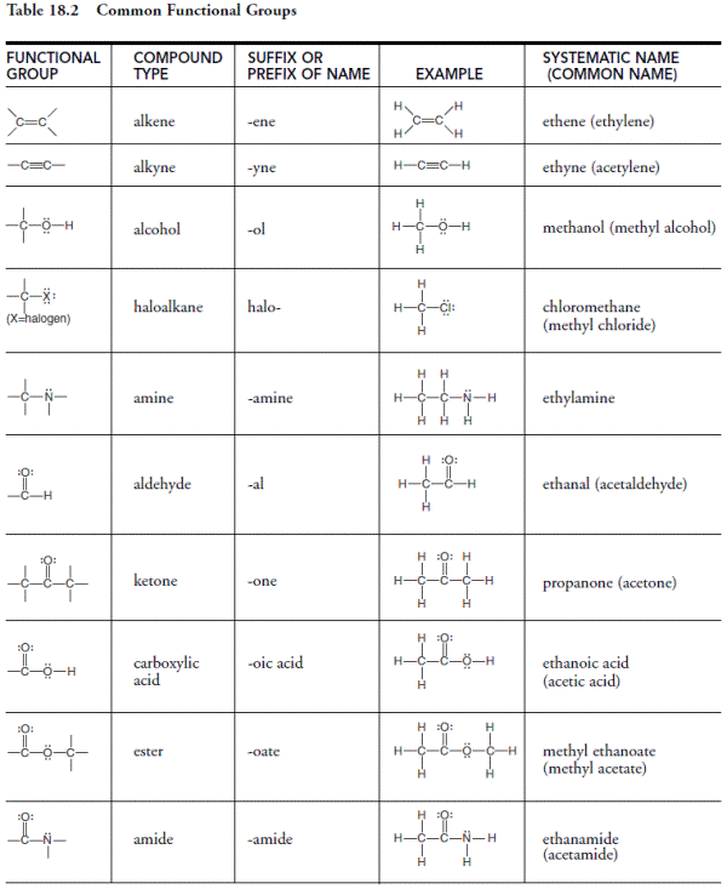
Alkanes, alkenes, and alkynes are merely the backbone of the majority of the compounds found on our Earth. Attached to the group are many different types of functional groups. There are a wide variety of functional groups, but the names and structures of the majority of the functional groups can be seen in the table below. NOTE - You should know the structure and name of all of these compounds:
Practice, Practice, Practice!
|
Before trying to take down the pros, make sure to practice your beach volleyball moves on the following practice problems:
|
The Final Match!
|
Once you feel that you are prepared to get your hair covered with sand, try your bump, set, and spike at this FRQ problem. NOTE - If you need to retake this FRQ, then you need to see Mr. Astor for a new problem.
|