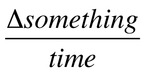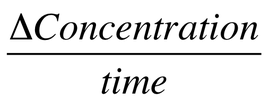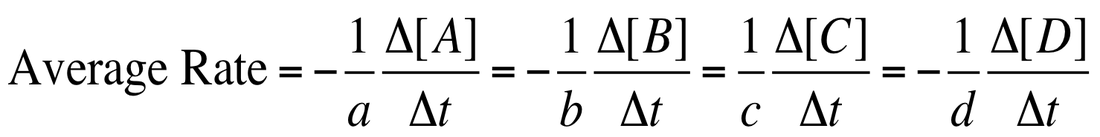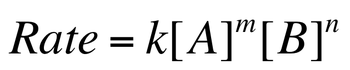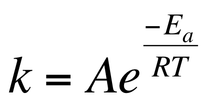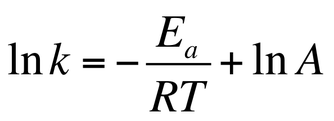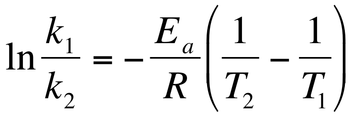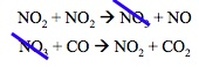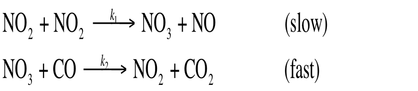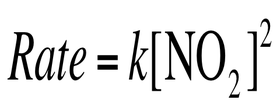Track and Field - Chemical Kinetics
|
Perhaps the most famous event from the Olympics, track and field is truly a sport that is amazing to watch. The speed at which they are able to run 100, 200, and 400 m is truly spectacular.
Chemical reactions also occur at an equally fast rate. In order to achieve at the highest levels and cross the finish line at the top of the medal stand, you will need to be able to complete questions concerning all of the topics are are listed below. |
Average Rates
When we describe the rate of any process, we mean:
When examining the rate of chemical reaction, we examine change in concentration over a period of time:
When we describe the rate of any process, we can think of the reaction:
aA + bB --> cC + dD
We can describe it as the rate of disappearance of A or B or the appearance of C or D:
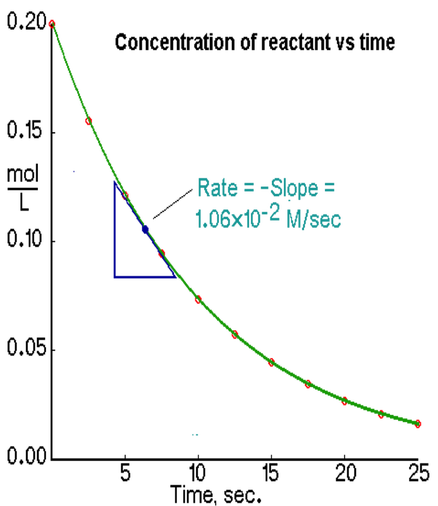
Graphing Average and Instantaneous Rates
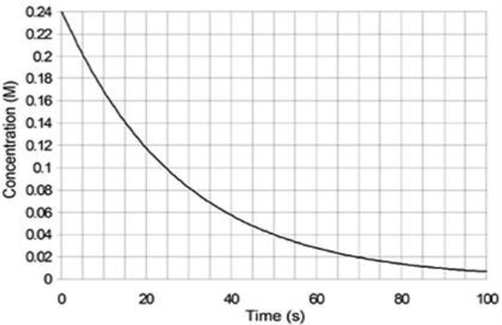
We can plot the concentration against time on an x/y coordinate plane. The line that is generated is known as the average rate and it general looks like:
While the average rate is useful, it has limitations because we cannot identify the rate at a given period of time. The graph on the last slide allows us to examine the instantaneous rate. The slope of the tangent line at a given point in time gives us the instantaneous rate. The slope of the tangent line usually looks like:
Rate Laws
The initial concentrations of the reactants allow for us to determine the rate of a reaction. The rate law shows the relationship between the reactant concentrations and the rate. It must be experimentally determined. For the general reaction:
a A + b B --> c C + d D
The rate law is
k = rate constant (temperature dependent)
m and n = whole numbers greater than 0
m and n = whole numbers greater than 0
Once m and n for the above reaction is determined, the reaction order is:
Because rate laws are experimentally determined, rate constants must also be determined experimentally. Once the exponents are known, one of the sets of data can be put in the rate law and k can be determined. Units
of k depend on the reaction order. The
units of k must cancel out concentration units so that the rate can be in units
of M x s-1
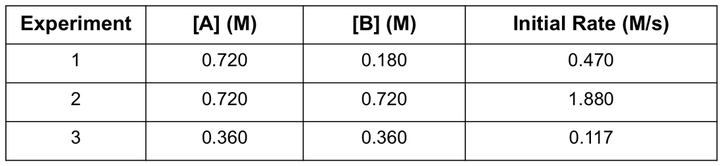
For example if we have the following data for the reaction previously described:
We can use this data by dividing rate law for experiment 1 by the rate law for experiment 2. As you can see, the concentrations of A and k will cancel out because they have identical values for experiment 1 and 2, so we can solve for the value of n. REMEMBER - in order to bring an exponent in front of a function, we need to take the logaritm of both sides. From this we can solve for n and then complete a second calculation to solve for m.
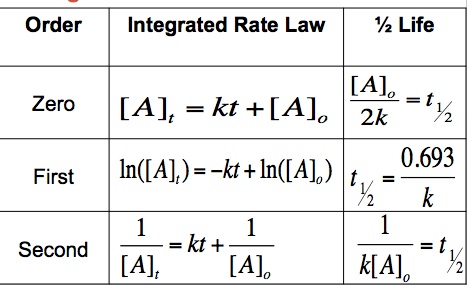
Integrated Rate Laws
Using calculus, we can integrate rate laws. This allows for us to calculate the concentration of a given reactant at a certain point in time. If we know the reaction order, then we have an equation that we can use to calculate the concentration of a given period of time as long as the time and initial concentration are known. The integrated rate laws for zero, first, and second order reactions can be seen below:
The ½ life of a reaction describes the amount it takes until only half of the initial concentration remains. The faster the reaction, the shorter the half – life
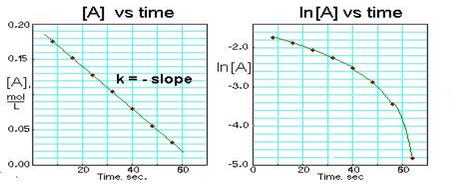
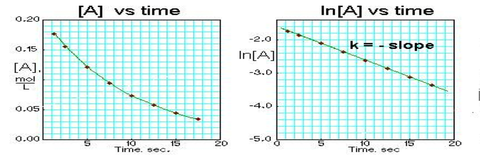
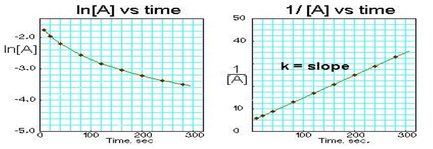
Since each of the integrated rate laws are in the form of linear equations, we can graph the concentration against time and the function that generates a linear curve indicates if our reaction is zero, first, and second order. You always need to place time along the x axis, but you can place either [A], ln([A]), or 1/[A] on the y axis. The y axis term that generates a linear plot will indicate the overall order of the reaction
Zero Order Graph
First Order Graph
Second Order Graph
Arrhenius Equation
Reaction rates can be altered by changing both temperature and concentration. Collision Theory is an application of kinetic molecular theory. The central idea of the theory is that collisions are what leads to chemical reactions. Greater the number of collisions = higher rate.
For collision theory, molecules must be oriented in a certain way during collision for a reaction to occur. Incorrect orientations will not lead to a reaction even if there are collisions
For collision theory, molecules must be oriented in a certain way during collision for a reaction to occur. Incorrect orientations will not lead to a reaction even if there are collisions
|
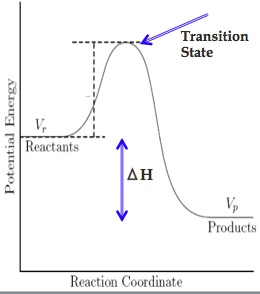
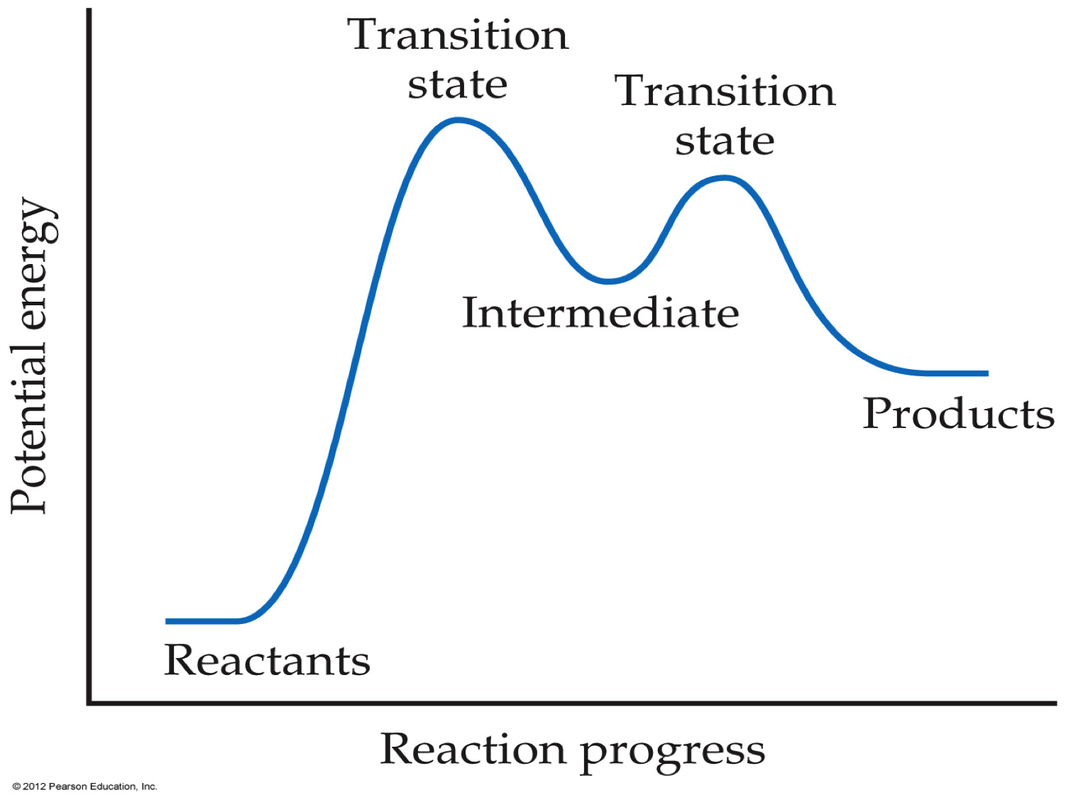
Molecules must possess a certain amount of energy to react. We use a reaction coordinate to help describe the progress of a reaction. The area of highest energy represents the transition state between reactants and products
Kinetic molecular theory explains why increasing temperature increases the rate of reaction. A greater number of molecules possess the necessary kinetic energy for a reaction to occur, this is why rate is increased. |
The rate constant for a reaction is temperature and activation energy dependent. The Arrhenius equation relates the rate constant to activation energy and temperature. The Arrhenius equation is:
This can be turned into a linear function through the equation:
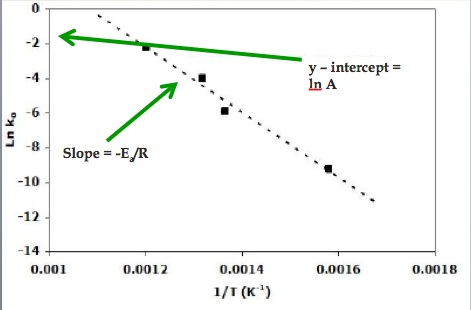
This is now in the form y = mx + b. We can graph ln k against the inverse of temperature. The slope is –Ea/T. The y – intercept is ln A
Finally, we can combine two Arrhenius equations at two different temperatures and it generates an equation that allows for us to calculate the new value of k if the temperature is changed so long as the the activation energy, both temperatures, and one equilibrium constant is known.
Reaction Mechanisms
The steps that a reaction takes in order to occur is the reaction mechanism. These are the steps of bonds breaking, atoms rearranging, and new bonds forming. Reactions that occur in a single step are elementary reactions. The number of molecules that participate in elementary
reactions is known as the molecularity of the reaction.
- Unimolecular – Single
molecule involved
- Bimolecular –
Collision of two molecules required
- Termolecular –
Collision of three molecules required
Most chemical reactions require multiple steps in order to occur. For example the reaction:
NO2 + CO --> NO + CO2
Requires 2 steps in order to occur:
|
They must create an intermediate, those molecules that are produces and then consumed before reaction completion, before producing the final product(s).
Every reaction is made up of one or more elementary reaction. If the reaction is elementary, then its rate law is based on molecularity. For example, if the reaction is elementary and it is of the form: A --> Products
Then the rate law is simply:
Rate = k [A]
|
The majority of reactions require 2 or more elementary reactions. Each step has its own rate constant and activation energy. The overall rate of the reaction can not exceed the rate of the slowest elementary reaction. The slow step is the rate – determining step. The rate – limiting step greatly inhibits mechanism and rate law.
For the multistep reaction previously discussed:
For the multistep reaction previously discussed:
The first step is the rate limiting step, so k2 >> k1. Therefore, we can only use the rate constant and reaction from the slow first step. Therefore, the rete law is:
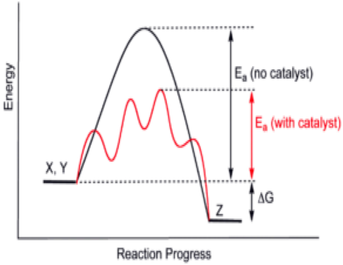
Catlaysts
|
A catalyst is a substance that speeds up a chemical reaction by lowering the activation energy. It provides an alterative pathway by which the reaction can occur. ΔG is not impacted.
A homogeneous catalyst is a catalyst whose phase is the same as the reactants. A heterogeneous catalyst is a catalyst whose phase is different from that of the reactants. |
Practice, Practice, Practice!
|
One does not become the world's fastest runner overnight. It takes years of training and commitment.
Train your brain with the following kinetics questions and check your answers when you are done: |
The Final Race!
|
Once you feel that you have adequately completed the training exercises. Try your hand at this sprint of a kinetics problem. NOTE - If you need to reatake the kinetics problems, then you need to see Mr. Astor for a new FRQ
|