Gymnastics - Quantum Theory and the Periodic Table
|
The first event of the 2013 AP Olympics is the agile sport of gymnastics. While you may begin firmly planted on the ground, there is a large amount of uncertainty about where you will end up once the event begins. In this event you will study the depths of quantum mechanics, the history behind the discoveries that were made and the extensive amounts of applications that can be seen in areas ranging from the Periodic Table to chemical bonding!
|
Quantum Theory and Calculations
The development of quantum theory can seem rather complex, but the following 6 videos provide a clear explanation of the complete development of the theory.
Quantum Numbers and Orbitals
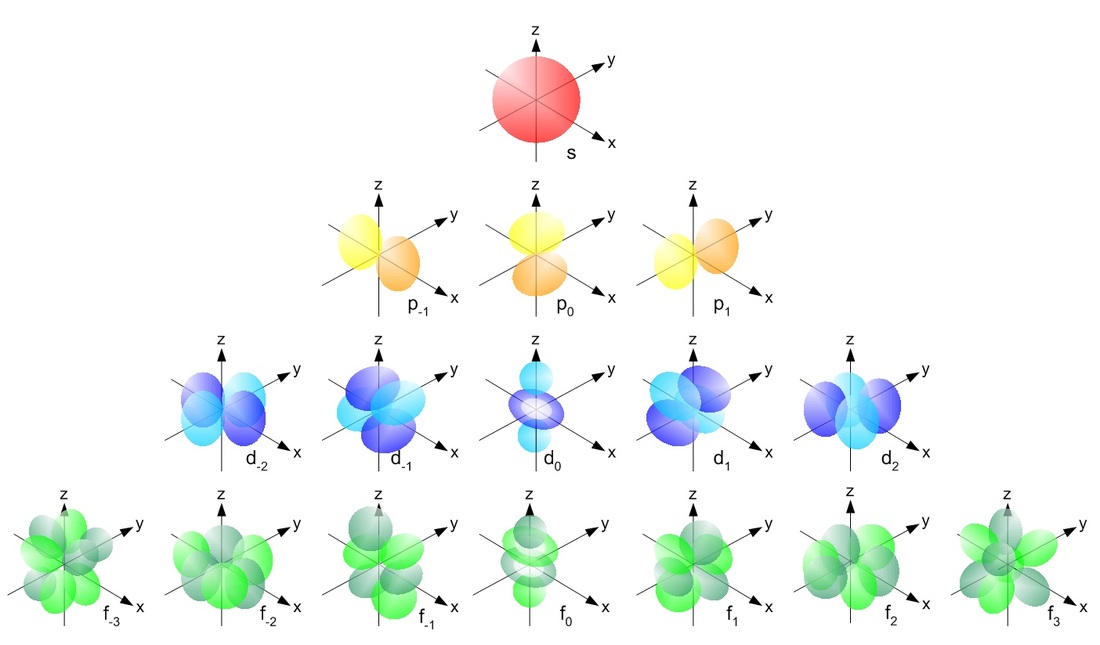
Because of Schroodinger's equation and the development of the quantum mechanical model, scientists were able to understand atom through the idea of orbitals and quantum numbers. If we think about an electron in an atom, we can describe it using four quantum numbers (n, l, ml, and ms). No two electrons in the same atom can have the same 4 quantum numbers. The quantum numbers can be described as:
- n = Principle Quantum Number: This is the energy level that that the electron occupies. n can be any number greater than or equal to 1.
- l = Azimuthal Quantum number. This value describes the shape of the probability density. It can have any value of 0 to n-1. The value indicates the shape. 0 is the s orbital, 1 is the p orbital, 2 is the d orbital, and 3 is the f orbital.
- ml = Magnetic Quantum Number: This is the orientation of the electron probability density along an x/y/z coordinate plane. This can have any value in the range of -l to +l. The sum of these represents the number of possible orientations that the electron probability density can take.
- ms = Angular Spin Number: This is the spin of the electron. It can have a value of either +1/2 or -1/2
Periodic Trends
Periodic trends emerge largely due to the effective nuclear charge that an atom possesses. Review your notes and watch the video below to remind yourself about what effective nuclear charge of an atom describes:
Because of effective nuclear charge, numerous trends are observed. We studied the following trends and their relationship to effective nuclear charge:
- Ionization Energy
- Electron Affinity
- Atomic and Ionic Radii
Lattice Energies
Lattice energies are the energy that is required to take ions in a crystal lattice and pull them apart until they are completely separated. The energy that is required depends upon two main factors:
1. The charge of the ions
2. The size of the ions
The larger the charge, the larger the energy needed to pull them apart and the smaller the ion, the larger the energy needed to pull the ions apart.
1. The charge of the ions
2. The size of the ions
The larger the charge, the larger the energy needed to pull them apart and the smaller the ion, the larger the energy needed to pull the ions apart.
Practice, Practice, Practice!
|
The following worksheets will help you practice synthesizing all of the information that will allow for you to become an expert quantum gymnast.
|
The Main Event!
|
When you feel you have adequately trained, then complete the FRQ problem below in order to medal in the event! NOTE - If you need to retake, then you need to see Mr. Astor for the other FRQ options.
|