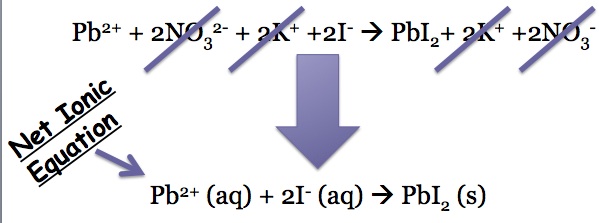Boxing - Writing Chemical Reactions
|
We have been hit in the face quite a few times by having to write chemical reactions and predict the problems that are formed. Despite all of these hits to our face, and possible ego, we need to get back up and punch back because this will be the basis of FRQ #4 on the exam.
This event is worth the maximum amount of points, so success on this event will indicate a level of preparedness that is unrivaled by other AP athletes. |
Writing Chemical Formulas
Being able to name and write inorganic compounds is essential to you landing that winning side hook. Watch the following video to help you remember how to write these formulas
Polyatomic Ions To MEMORIZE! No Exceptions!
Writing Net Ionic Equations
When you dissolve compounds in water, sometimes they dissociate and sometimes they do not.An electrolyte is any compound that breaks into ions when in solution and can conduct electricity. A nonelectrolyte is any compound that does not form ions when in solution and cannot conduct electricity.
When a ionic compound is dissolved in water, it breaks apart into positive and negative ions. Polar water molecule breaks up the ionic crystal. When a covalent compound is dissolved in
water, it cannot be broken apart. Polar water molecule
cannot break apart these compounds because there are no ions.
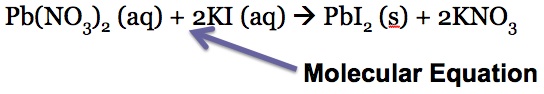
Oftentimes we see chemical equations written as this:
Oftentimes we see chemical equations written as this:
The electrolytes that are in aqueous solutions can be written as their ions:
Many of the ions are present on both sides, so they do not participate in the chemical reaction and can be canceled out when the net ionic equation is written:
Types of Reactions
The main types of chemical reactions that you will be assessed on are as follows:
1. Synthesis
2. Decomposition
3. Single Replacement
4. Double Replacement
5. Precipitation
6. Neutralization
7. Oxidation-Reduction
8. Complex Ions
9. Simple Organic Problems
Every year, free response question 4 requires you to predict the products that form for three chemical reactions. Therefore, being able to predict chemical reactions is vitally important to your overall success on the exam. While the following video is long, it provides an extensive overview on how to predict these reactions:
1. Synthesis
2. Decomposition
3. Single Replacement
4. Double Replacement
5. Precipitation
6. Neutralization
7. Oxidation-Reduction
8. Complex Ions
9. Simple Organic Problems
Every year, free response question 4 requires you to predict the products that form for three chemical reactions. Therefore, being able to predict chemical reactions is vitally important to your overall success on the exam. While the following video is long, it provides an extensive overview on how to predict these reactions:
Redox Reactions and Oxidation Numbers
Oxidation-Reduction reactions or redox reactions are reactions that involve the transfer of electrons. These are one of the most common chemical reactions. In order to identify redox reactions, you need to be able to label oxidation states of elements. Watch the video below to review how to do this.
In addition to oxidation states, redox reactions can be predicted using an activity series. Remember you are being given a reduction potential table on the exam. This can help you predict which reactions run in the forward reaction and which reactions run in the reverse direction! Check out the simulator below to visualize this:
Complex Ion Formation
Transition metals are known to form complex ions when a ligand is added to solution. A ligand is any substance that can be classified as a Lewis base (it donates electrons to solution). While complex ion reactions are not seen often in the equations section, it is helpful to know a few common components that are typical of these complexes.
The most common complex ion metals (the Lewis acid) are:
The most common ligands (the Lewis base) are:
While they are seen infrequently, it is suggested that you attempt a few problems in order to be prepared just in case!
The most common complex ion metals (the Lewis acid) are:
- Fe, Co, Ni, Cr, Cu, Zn, Ag, and Al
The most common ligands (the Lewis base) are:
- NH3, CN-, OH-, and SCN-
While they are seen infrequently, it is suggested that you attempt a few problems in order to be prepared just in case!
- Complex Ion Practice (p. 87)
Naming Basic Organic Compounds
Recall, a hydrocarbon is a molecule that contains only hydrogen and carbon. Most often, the molecule is a carbon backbond with the additional spaces filled by hydrogen. If the chain only has single bonds, then we call it an alkane. An alkene is a hydrocarbon chain with at least 1 double bond. An alkyne is a hydrocarbon chain with at least 1 triple bond.
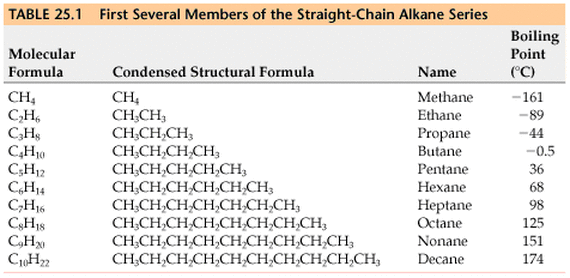
We have a systematic way by which we can name alkanes. Based on the number of carbons on the longest hydrocarbon chain, we attach a prefix that ends with the root -ane in order to signify the number of carbons. The roots for 1 - 10 carbon long chains are seen in the table below:
We have a systematic way by which we can name alkanes. Based on the number of carbons on the longest hydrocarbon chain, we attach a prefix that ends with the root -ane in order to signify the number of carbons. The roots for 1 - 10 carbon long chains are seen in the table below:
If the longest hydrocarbon chain contains a double bond, then the -ane suffix is replaced by -ene (i.e. propene, butene, pentene, etc). If the longest hydrocarbon chain contains a triple bond, then the -ane suffix is replaced with -yne (i.e. butyne, pentyne, hexyne, etc.).
Predicting Reactions
When predicting a chemical reaction for FRQ #4, here are the steps that you should follow:
1. What are the compounds that are given in the question?
1. What are the compounds that are given in the question?
- You get 1 point just for writing the reactants. What are the compounds or elements that are given? Write both of these down in order to enhance your chance of success?
- Is it an "easy to spot" type of equation? This could be a decomposition, synthesis, or single replacement. Each of these are fairly easy to spot. Decomposition is when you start with only one compound and it has to split apart. Synthesis is easy to see when you have 2 elements to begin with (the only option for a reaction is to push the two elements into one new compound). Finally, single replacement is easy to spot when you have one compound and one element. It is likely that the element will replace one of the elements in the compound of interest.
- Could 1 element replace another in the compound to make a solid? REMEMBER THE SOLUBILITY RULES!
- Does it contain things like NO3, SO3, or CO3, which will turn into gases like NO2, SO2, or CO2 upon heating or the addition of compounds like acids?
- Is it a reaction between one of the strong acid and one of the strong bases? Then it is a neutralization reactions!
- Unsure? It may be one of the more uncommon types of reactions such as complex ion formation and/or organic chemistry problems. Use your best judgement to solve this problem!
- Using the rules that were previously discussed, breakdown the molecular equation to the complete ionic, which can be translated to the net ionic equation.
Practice, Practice, Practice!
|
Once you feel you have mastered the content, try your right hook at these practice problems!
|
The Main Event!
|
LET'S GET READY TO RUMBLE! Time to take down FRQ #4. Put your game face on and take on this problem. NOTE - If you need to retake this FRQ, then you need to see Mr. Astor for a new FRQ problem.
|