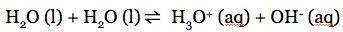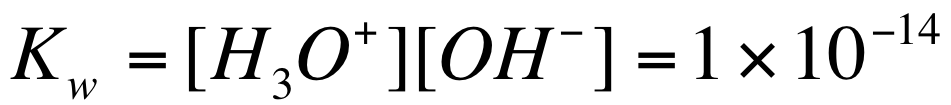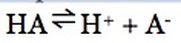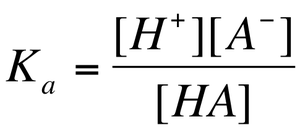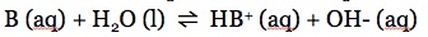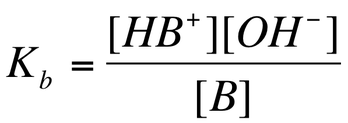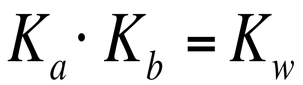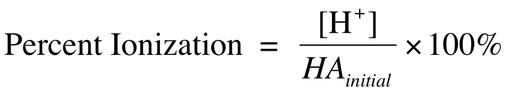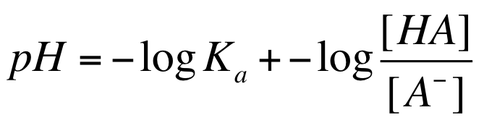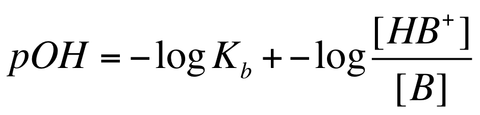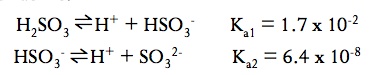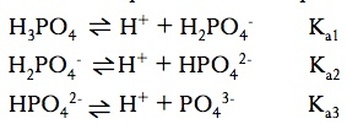Basketball - Acids and Bases
|
Basketball is largely a sport that has been dominated by the USA over the past olympic events, but with the competitive field that has been emerging, it is really anyone's game in the upcoming olympics.
In order to be victorious on the basketball court you are going to need to master the basics of acid/base chemistry, calculating pH for weak acid/base equilibrium, titration curves, and buffers. |
Acid and Base Gen. Chem Review
Recall, one of the most common types of reactions are acid/base reactions or neutralization reactions. We have various definitions of acids and bases, but they all react with characteristic properties.
Arrhenius’ theory of acids/bases focuses on what molecules contain. An Arrhenius acid contains a H+ and an Arrhenius base contains an OH-.
While Arrhenius’ definition covers many acids/bases, acid/base reactions are not about containing H+ or OH-. A Bronsted-Lowry acid is any compound that gives H+ during the reaction. A Bronsted-Lowry base is any compound that accepts H+ during the reaction.
Ultimately, acid/base reactions are not about H+, but about the accepting or donating of electron pairs. A Lewis acid is an electron pair acceptor and a Lewis base is an electron pair donor. Every acid/base in the previous two categories are also acids/bases in this category. This is the broadest definition of acids/bases.
Arrhenius’ theory of acids/bases focuses on what molecules contain. An Arrhenius acid contains a H+ and an Arrhenius base contains an OH-.
While Arrhenius’ definition covers many acids/bases, acid/base reactions are not about containing H+ or OH-. A Bronsted-Lowry acid is any compound that gives H+ during the reaction. A Bronsted-Lowry base is any compound that accepts H+ during the reaction.
Ultimately, acid/base reactions are not about H+, but about the accepting or donating of electron pairs. A Lewis acid is an electron pair acceptor and a Lewis base is an electron pair donor. Every acid/base in the previous two categories are also acids/bases in this category. This is the broadest definition of acids/bases.
|
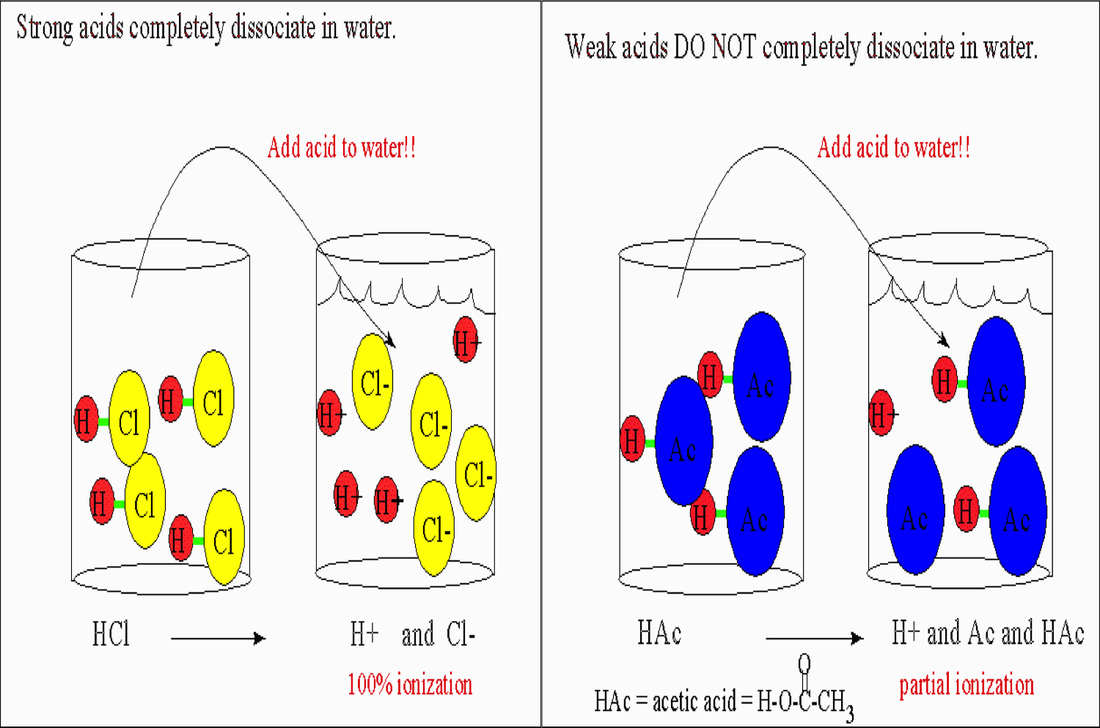
Acids/Bases can be classified as strong or weak based on how much it disassociates. To disassociate means to break apart. Acids/Bases that completely dissociate are called strong acids/bases. Acids/Bases that only partially dissociate are called weak acids/bases. These are in equilibrium with one another
The seven strong acids are:
1. HCl 2. HBr 3. HI 4. HNO3 5. H2SO4 6. HClO3 7. HClO4 The eight strong bases are: 1. LiOH 2. NaOH 3. KOH 4. RbOH 5. CsOH 6. Ca(OH)2 7. Sr(OH)2 8. Ba(OH)2 |
The concentrations of acids and bases are often very low. We use the pH scale to convey the concentration of H+. The pH scale is 0-14. An acid is anything with a pH 0-7, a base is anything with a pH 7-14, and neutral chemicals are substances with a pH of 7. We calculate the pH by:
pH = -log [H+]
We calculate the pOH by:
pH = -log [OH-]
For strong acids and bases, the concentration of the compound is the [H+] or [OH-]. For bases, we only know [OH-], so we need to first calculate pOH and then convert to pH using the equation:
pH = 14 - pOH
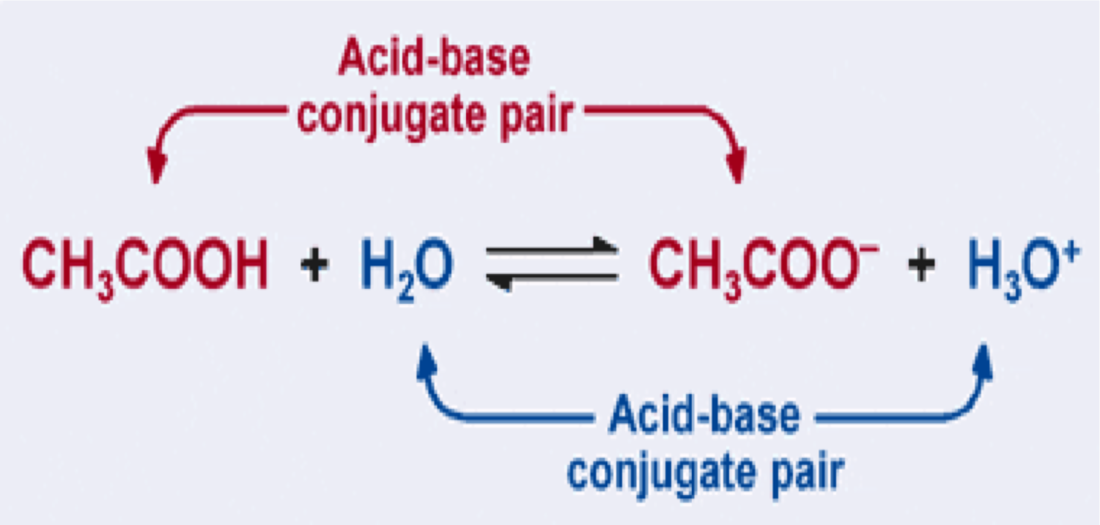
—Every acid has a conjugate base that is formed when a proton (H+) is removed. Every base has a conjugate acid that forms when a proton is added
Strong Acid/Strong Base Titrations
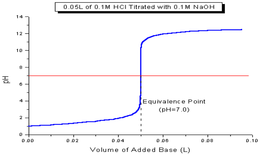
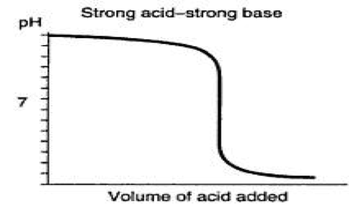
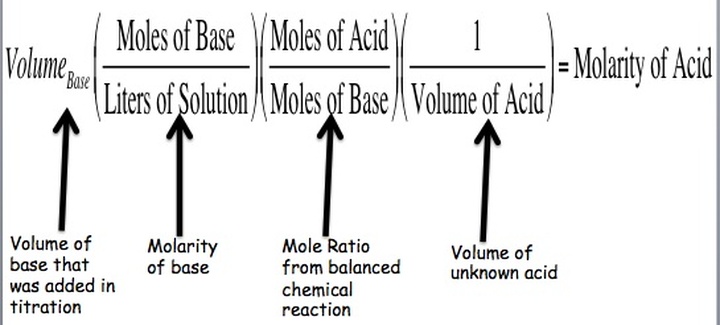
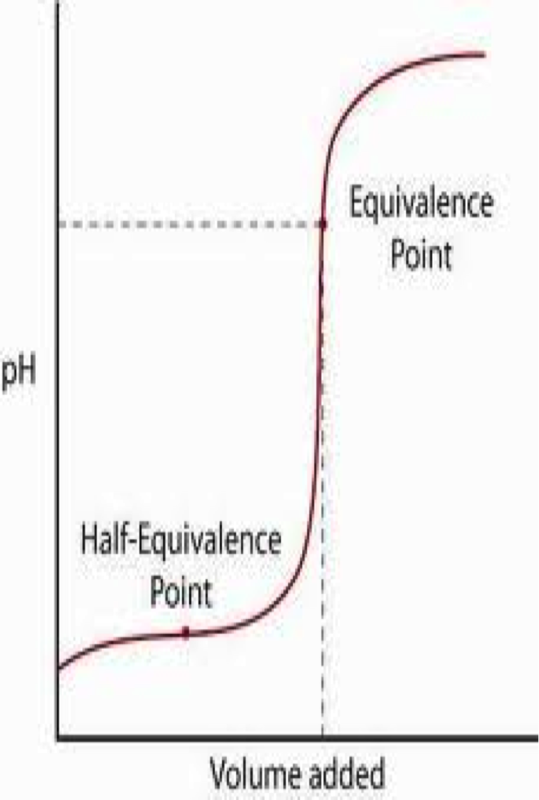
Often, we have solutions of unknown concentrations. A titration is the controlled addition of solution of known concentration/volume to a solution of known volume, but unknown concentration. We use a standard solution during titrations, which is a solution whose concentration and volume with know precisely. We can graph the pH of a titration solution against the volume of amount added and we create a titration curve. The two strong acid/base titration curves can be seen below.
Ultimately, we are trying to spot the equivalence point for the the titration. The equivalence point is the point at which the two solutions used in a titration are present in equal amounts. We use indicators that change within a narrow pH range in order to spot this point. For strong acid/strong base titrations, it occurs at pH = 7. At this point, the moles of acid are equal to the moles of base. This means that we can convert between acid and base concentrations using the formula:
Weak Acid/Base Equilibrium
Water can act as either an acid or a base. It is amphoteric, it can function as either an acid or a base. Therefore, in a solution of water, we have the equilibrium of:
We can calculate the equilibrium constant of this reaction to be:
Recall, a weak acid is partially dissociated. Therefore, reactants and products are in equilibrium:
The equilibrium constant can be written for all weak acids as:
The equilibrium constant for a weak acid equilibrium is known as the acid dissociation constant (Ka).
Just like with weak acids, a weak acid partially dissociates. Therefore, reactants and products are in equilibrium:
Just like with weak acids, a weak acid partially dissociates. Therefore, reactants and products are in equilibrium:
The equilibrium constant for a weak base equilibrium is known as the base dissociation constant (Kb) and it can be written as:
Because a weak acid is simply the reverse of a conjugate base reaction (and vice versa for the base), we can relate Ka to Kb through the following equation:
When working with a weak acid or weak base, we cannot simply use the concentration of solution to calculate pH because there is an equilibrium. Therefore, we need to do an ICE table in order to calculate the pH of the solution. This ICE table is just like the ones that we completed in the swimming event. We know the initial concentration of either the weak acid or weak base and we can use these concentration to determine the values at equilibrium by adding or subtracting the appropriate amount of x (See video for more detailed example).
The size of Ka indicates the strength of an acid (the same for Kb). The larger the value of Ka, the stronger the acid. Another way to measure strength is the percent ionization. This can be calculated through the equation:
The size of Ka indicates the strength of an acid (the same for Kb). The larger the value of Ka, the stronger the acid. Another way to measure strength is the percent ionization. This can be calculated through the equation:
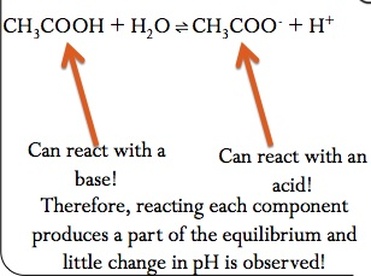
Buffers
A buffer is any solution that resists changes in pH. There are —two components of a buffer, which are:
1. A component that neutralizes an acid
2. A component that neutralizes a base
A weak acid or a weak base are capable of creating a buffer because they have both of these components, but a strong acid or base cannot create a buffer.
A weak acid or base make an excellent buffer because they have a component that can react with an acid and a component that can react with a base.
1. A component that neutralizes an acid
2. A component that neutralizes a base
A weak acid or a weak base are capable of creating a buffer because they have both of these components, but a strong acid or base cannot create a buffer.
A weak acid or base make an excellent buffer because they have a component that can react with an acid and a component that can react with a base.
The best buffers have close to equal concentrations of the conjugate acid/base pair. This is due to the fact that the ratio of acid to conjugate base is what determines the concentration of H+ that is observed at equilibrium. The closer this ratio is to 1, the more likely that the pH will see minimal change when an external stress is introduced.
When we have a buffer, we do not need to use ICE tables to determine the pH of the solution. We can use the Henderson-Hasselbalch equation in order to solve for the pH. The HH equation for weak acids is:
When we have a buffer, we do not need to use ICE tables to determine the pH of the solution. We can use the Henderson-Hasselbalch equation in order to solve for the pH. The HH equation for weak acids is:
Furthermore, we can write the HH equation for a weak base/conjugate acid buffer by utilizing the following equation:
Weak Acid/Base Titrations
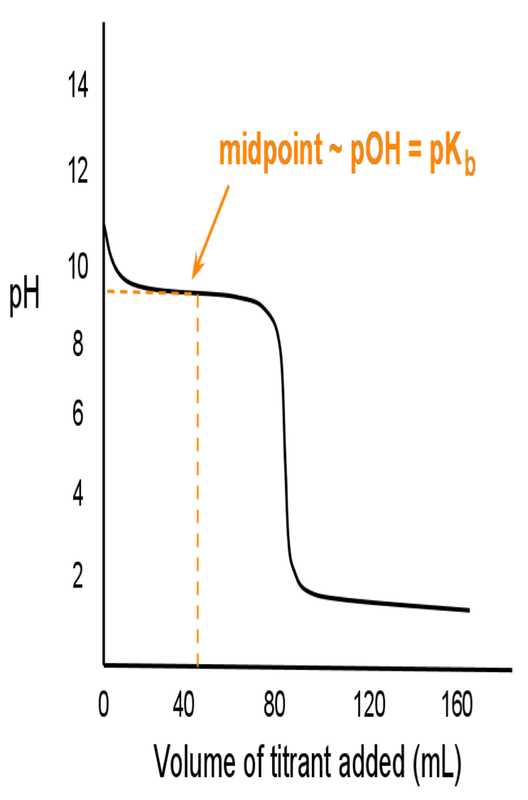
When we titrate a weak acid/base with a strong acid/base, the titration curve looks different than the ones previously studied. A strong acid/base titration goes to completion the instant we add the solution. —A weak acid/base titration reacts and than reestablishes equilibrium.
Therefore, we need to do two steps upon the introduction of a strong acid/base to a weak acid/base equilibrium:
1. React the strong acid or base to completion with the weak acid/base of interest. This means that we need to use a BCA table in order to determine the amount of moles of acid/conjugate base that are present once the reaction has gone to completion.
2. Reestablish equilibrium by completing an ICE table by utilizing the new moles to calculate the new initial molarity. From this point, we can calculate the new equilibrium concentration using the ICE table that we have become accustomed to at this point.
The titration curves for a weak acid titrated with a strong base or a weak base titrated with a strong acid are as follows (note that the equivalence point is no longer at 7 due to the presence of conjugate acid or base at equilibrium):
Therefore, we need to do two steps upon the introduction of a strong acid/base to a weak acid/base equilibrium:
1. React the strong acid or base to completion with the weak acid/base of interest. This means that we need to use a BCA table in order to determine the amount of moles of acid/conjugate base that are present once the reaction has gone to completion.
2. Reestablish equilibrium by completing an ICE table by utilizing the new moles to calculate the new initial molarity. From this point, we can calculate the new equilibrium concentration using the ICE table that we have become accustomed to at this point.
The titration curves for a weak acid titrated with a strong base or a weak base titrated with a strong acid are as follows (note that the equivalence point is no longer at 7 due to the presence of conjugate acid or base at equilibrium):
Polyatomic Acids/Bases
Polyprotic acids are acids have more than one H that can be considered acidic. For example, H2SO3 has two Ka values due to the fact that it has two acidic protons
Each removal of a proton has it’s own Ka value. It is always easier to remove the first proton from a polyprotic than it is is to remove the second proton. As long as Ka1 > Ka2 by a factor of at least 1000, then we only need to consider first Ka when calculating values like pH.
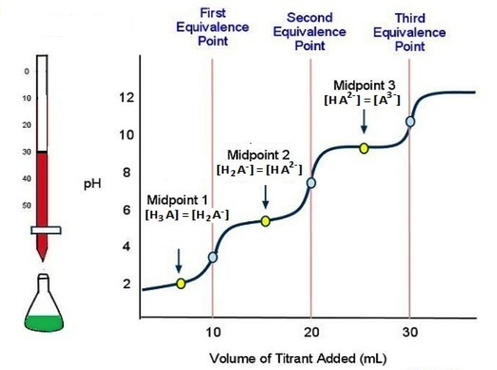
When titrating a polyprotic acid with a strong base, the reaction proceeds in multiple steps.
When titrating a polyprotic acid with a strong base, the reaction proceeds in multiple steps.
All of the first species must complete react before the second proton is removed. —There will be the same number of equivalence points as there are acidic protons.
Practice, Practice, Practice!
|
Once you feel you have adequately covered all of the content for acids and bases, try your hand with a little practice on the court before going up against the pros. Make sure to check your answers when you are finished!
|
The Championship Game!
|
Think you have what it takes to takedown Kobe? Once you feel that you have practiced enough to take on the pros, complete the FRQ problem that is linked below. NOTE - If you need to retake this event, then you need to get a new problem from Mr. Astor
|